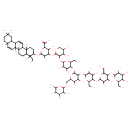
Identification Common Name Phaseoloside E Class Small Molecule Description Phaseoloside E is found in pulses. Phaseoloside E is isolated from Phaseolus vulgaris (kidney bean). Contaminant Sources Contaminant Type Not Available Chemical Structure Synonyms Value Source Air edale blue 2BD HMDB Airedale blue 2BD HMDB Aizen direct blue 2BH HMDB Amanil blue 2BX HMDB Atlantic blue 2b HMDB Atul direct blue 2b HMDB Azocard blue 2b HMDB Azomine blue 2b HMDB Belamine blue 2b HMDB Bencidal blue 2b HMDB Benzanil blue 2b HMDB benzo Blue bba-CF HMDB benzo Blue BBN-CF HMDB benzo Blue GS HMDB Blue 2b HMDB Blue 2b salt HMDB Brasilamina blue 2b HMDB Brazilamina blue 2b HMDB C.I. dir ect blue 6, tetrasodium salt HMDB C.I. direct blue 6 HMDB C.I. direct blue 6, tetrasodium salt HMDB Calcomine blue 2b HMDB Chloramine blue 2b HMDB Chlorazol blue b HMDB Chlorazol blue BP HMDB Chrome leather blue 2b HMDB Cresotine blue 2b HMDB Diacotton blue BB HMDB Diamine blue HMDB Diamine blue 2b HMDB Diamine blue BB HMDB Diaphtamine blue BB HMDB Diazine blu e 2b HMDB Diazine blue 2b HMDB Diazol blue 2b HMDB Diphenyl blue 2b HMDB Diphenyl blue KF HMDB Diphenyl blue m2b HMDB Direct blue HMDB Direct blue 2b HMDB Direct blue 2ba HMDB Direct blue 6 HMDB Direct blue 6 (technical grade) HMDB Direct blue a HMDB Direct blue BB HMDB Direct blue GS HMDB Direct blue K HMDB Direct blue m2b HMDB Direct sky blue K HMDB Enianil blue 2BN HMDB Fenamin blue 2b HMDB Fixanol blue 2b HMDB Hispamin blue 2b HMDB indigo Blue 2b HMDB Kayaku direct HMDB Kayaku direct blue BB HMDB Mitsui direct blue 2BN HMDB Naphtamine blue 2b HMDB Niagara blue 2b HMDB Nippon blue BB HMDB Paramine blue 2b HMDB Phenamine blue BB HMDB pheno Blue 2b HMDB Pontamine blue BB HMDB Tertrodirect blue 2b HMDB Vondacel blu e 2b HMDB Vondacel blue 2b HMDB 4-[(3-{[5-({3-[(5-{[3,4-dihydroxy-6-(hydroxymethyl)-5-{[3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}oxan-2-yl]oxy}-3,4-dihydroxy-6-(hydroxymethyl)oxan-2-yl)oxy]-4,5-dihydroxy-6-{[(3,4,5-trihydroxy-6-methyloxan-2-yl)oxy]methyl}oxan-2-yl}oxy)-3,4-dihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}-4,5-dihydroxyoxan-2-yl)oxy]-3,5-dihydroxy-6-{[4-(hydroxymethyl)-4,6a,6b,8a,11,11,14b-heptamethyl-1,2,3,4,4a,5,6,6a,6b,8a,9,10,11,12,12a,12b,14a,14b-octadecahydropicen-3-yl]oxy}oxane-2-carboxylate Generator Blue, diamine MeSH Blue, trypan MeSH Niagara blue MeSH Blue, niagara MeSH Trypan blue MeSH VisionBlue MeSH
Chemical Formula C77 H124 O41 Average Molecular Mass 1705.784 g/mol Monoisotopic Mass 1704.762 g/mol CAS Registry Number 30915-09-4 IUPAC Name 4-[(3-{[5-({3-[(5-{[3,4-dihydroxy-6-(hydroxymethyl)-5-{[3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}oxan-2-yl]oxy}-3,4-dihydroxy-6-(hydroxymethyl)oxan-2-yl)oxy]-4,5-dihydroxy-6-{[(3,4,5-trihydroxy-6-methyloxan-2-yl)oxy]methyl}oxan-2-yl}oxy)-3,4-dihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}-4,5-dihydroxyoxan-2-yl)oxy]-3,5-dihydroxy-6-{[4-(hydroxymethyl)-4,6a,6b,8a,11,11,14b-heptamethyl-1,2,3,4,4a,5,6,6a,6b,8a,9,10,11,12,12a,12b,14a,14b-octadecahydropicen-3-yl]oxy}oxane-2-carboxylic acid Traditional Name 4-[(3-{[5-({3-[(5-{[3,4-dihydroxy-6-(hydroxymethyl)-5-{[3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}oxan-2-yl]oxy}-3,4-dihydroxy-6-(hydroxymethyl)oxan-2-yl)oxy]-4,5-dihydroxy-6-{[(3,4,5-trihydroxy-6-methyloxan-2-yl)oxy]methyl}oxan-2-yl}oxy)-3,4-dihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}-4,5-dihydroxyoxan-2-yl)oxy]-3,5-dihydroxy-6-{[4-(hydroxymethyl)-4,6a,6b,8a,11,11,14b-heptamethyl-1,2,3,4a,5,6,9,10,12,12a,12b,14a-dodecahydropicen-3-yl]oxy}oxane-2-carboxylic acid SMILES CC1OC(OCC2OC(OC3C(O)C(O)C(OC4C(O)C(O)COC4OC4C(O)C(OC5CCC6(C)C(CCC7(C)C6C=CC6C8CC(C)(C)CCC8(C)C=CC76C)C5(C)CO)OC(C4O)C(O)=O)OC3CO)C(OC3OC(CO)C(OC4OC(CO)C(OC5OC(CO)C(O)C(O)C5O)C(O)C4O)C(O)C3O)C(O)C2O)C(O)C(O)C1O InChI Identifier InChI=1S/C77H124O41/c1-27-39(84)43(88)49(94)64(105-27)104-25-35-42(87)45(90)62(118-68-52(97)47(92)57(33(22-80)108-68)113-66-51(96)46(91)56(32(21-79)107-66)112-65-50(95)44(89)41(86)31(20-78)106-65)71(110-35)114-58-34(23-81)109-67(53(98)48(58)93)117-61-40(85)30(83)24-103-70(61)115-59-54(99)60(63(101)102)116-69(55(59)100)111-38-12-13-74(5)36(75(38,6)26-82)11-14-77(8)37(74)10-9-28-29-19-72(2,3)15-16-73(29,4)17-18-76(28,77)7/h9-10,17-18,27-62,64-71,78-100H,11-16,19-26H2,1-8H3,(H,101,102) InChI Key PZPHULADDDWLGC-UHFFFAOYSA-N