| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 03:30:51 UTC |
|---|
| Update Date | 2016-11-09 01:20:44 UTC |
|---|
| Accession Number | CHEM032574 |
|---|
| Identification |
|---|
| Common Name | Castanin |
|---|
| Class | Small Molecule |
|---|
| Description | Not Available |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
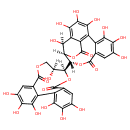
| Chemical Structure | |
|---|
| Synonyms | Not Available |
|---|
| Chemical Formula | C34H24O22 |
|---|
| Average Molecular Mass | 784.544 g/mol |
|---|
| Monoisotopic Mass | 784.076 g/mol |
|---|
| CAS Registry Number | 115406-24-1 |
|---|
| IUPAC Name | (14S,15R,19S)-14-[(10S,11S)-3,4,5,11,17,18,19-heptahydroxy-8,14-dioxo-9,13-dioxatricyclo[13.4.0.0²,⁷]nonadeca-1(19),2,4,6,15,17-hexaen-10-yl]-2,3,4,7,8,9,19-heptahydroxy-13,16-dioxatetracyclo[13.3.1.0⁵,¹⁸.0⁶,¹¹]nonadeca-1,3,5(18),6,8,10-hexaene-12,17-dione |
|---|
| Traditional Name | (14S,15R,19S)-14-[(10S,11S)-3,4,5,11,17,18,19-heptahydroxy-8,14-dioxo-9,13-dioxatricyclo[13.4.0.0²,⁷]nonadeca-1(19),2,4,6,15,17-hexaen-10-yl]-2,3,4,7,8,9,19-heptahydroxy-13,16-dioxatetracyclo[13.3.1.0⁵,¹⁸.0⁶,¹¹]nonadeca-1,3,5(18),6,8,10-hexaene-12,17-dione |
|---|
| SMILES | [H][C@]1(O)C2=C(O)C(O)=C(O)C3=C2C(=O)O[C@@]1([H])[C@]([H])(OC(=O)C1=CC(O)=C(O)C(O)=C31)[C@@]1([H])OC(=O)C2=CC(O)=C(O)C(O)=C2C2=C(O)C(O)=C(O)C=C2C(=O)OC[C@]1([H])O |
|---|
| InChI Identifier | InChI=1S/C34H24O22/c35-8-1-5-12(21(42)18(8)39)13-6(2-9(36)19(40)22(13)43)32(50)54-28(11(38)4-53-31(5)49)30-29-26(47)17-16(34(52)55-29)15(24(45)27(48)25(17)46)14-7(33(51)56-30)3-10(37)20(41)23(14)44/h1-3,11,26,28-30,35-48H,4H2/t11-,26-,28-,29+,30+/m0/s1 |
|---|
| InChI Key | CHBITXAMNKHJCR-KIJLVVMGSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as hydrolyzable tannins. These are tannins with a structure characterized by either of the following models. In model 1, the structure contains galloyl units (in some cases, shikimic acid units) that are linked to diverse polyol carbohydrate-, catechin-, or triterpenoid units. In model 2, contains at least two galloyl units C-C coupled to each other, and do not contain a glycosidically linked catechin unit. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Phenylpropanoids and polyketides |
|---|
| Class | Tannins |
|---|
| Sub Class | Hydrolyzable tannins |
|---|
| Direct Parent | Hydrolyzable tannins |
|---|
| Alternative Parents | |
|---|
| Substituents | - Hydrolyzable tannin
- Tetracarboxylic acid or derivatives
- Gallic acid or derivatives
- 2-benzopyran
- Isochromane
- Benzopyran
- 1-hydroxy-4-unsubstituted benzenoid
- 1-hydroxy-2-unsubstituted benzenoid
- Benzenoid
- Secondary alcohol
- Lactone
- Carboxylic acid ester
- Oxacycle
- Organoheterocyclic compound
- Polyol
- Carboxylic acid derivative
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Alcohol
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00kr-0006300900-a04afdf7c392009e6911 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-066v-0033000900-15007bc5be1845a2941b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0f92-0397800000-2b19bf14ee3dd627580d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-001l-0007000900-7bb4505b5f4c8f013868 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00lf-0005201900-e542bbbc656be6022b35 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0006-9445100300-b83df10e2d765e050d83 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | Not Available |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|