| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 03:24:20 UTC |
|---|
| Update Date | 2016-11-09 01:20:43 UTC |
|---|
| Accession Number | CHEM032457 |
|---|
| Identification |
|---|
| Common Name | Wybutoxine |
|---|
| Class | Small Molecule |
|---|
| Description | Wybutoxine is found in animal foods. Wybutoxine is a constituent of phenylalanine tRNA of beef, chicken, calf and rat livers. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
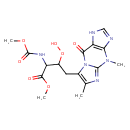
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Methyl 4,9-dihydro-b-hydroperoxy-a-[(methoxycarbonyl)amino]-4,6-dimethyl-9-oxo-1H-imidazo[1,2-a]purine-7-butanoate, 9ci | HMDB | | Peroxy-y-base | HMDB | | Peroxywybutine | HMDB | | Methyl 4-{4,6-dimethyl-9-oxo-3H,4H,9H-imidazo[1,2-a]purin-7-yl}-3-hydroperoxy-2-{[hydroxy(methoxy)methylidene]amino}butanoic acid | Generator |
|
|---|
| Chemical Formula | C16H20N6O7 |
|---|
| Average Molecular Mass | 408.366 g/mol |
|---|
| Monoisotopic Mass | 408.139 g/mol |
|---|
| CAS Registry Number | 36238-44-5 |
|---|
| IUPAC Name | methyl 4-{4,6-dimethyl-9-oxo-1H,4H,9H-imidazo[1,2-a]purin-7-yl}-3-hydroperoxy-2-[(methoxycarbonyl)amino]butanoate |
|---|
| Traditional Name | methyl 4-{4,6-dimethyl-9-oxo-1H-imidazo[1,2-a]purin-7-yl}-3-hydroperoxy-2-[(methoxycarbonyl)amino]butanoate |
|---|
| SMILES | COC(=O)NC(C(CC1=C(C)N=C2N1C(=O)C1=C(N=CN1)N2C)OO)C(=O)OC |
|---|
| InChI Identifier | InChI=1S/C16H20N6O7/c1-7-8(5-9(29-26)10(14(24)27-3)20-16(25)28-4)22-13(23)11-12(18-6-17-11)21(2)15(22)19-7/h6,9-10,26H,5H2,1-4H3,(H,17,18)(H,20,25) |
|---|
| InChI Key | FRYWGCQTDZARGT-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as alpha amino acid esters. These are ester derivatives of alpha amino acids. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Carboxylic acids and derivatives |
|---|
| Sub Class | Amino acids, peptides, and analogues |
|---|
| Direct Parent | Alpha amino acid esters |
|---|
| Alternative Parents | |
|---|
| Substituents | - Alpha-amino acid ester
- 6-oxopurine
- Hypoxanthine
- Imidazopyrimidine
- Purine
- Imidazo[1,2-a]pyrimidine
- Pyrimidone
- Fatty acid ester
- N-substituted imidazole
- Fatty acyl
- Pyrimidine
- Methylcarbamate
- Azole
- Heteroaromatic compound
- Imidazole
- Vinylogous amide
- Methyl ester
- Carbamic acid ester
- Lactam
- Carbonic acid derivative
- Hydroperoxide
- Carboxylic acid ester
- Alkyl hydroperoxide
- Monocarboxylic acid or derivatives
- Organoheterocyclic compound
- Azacycle
- Peroxol
- Organic oxide
- Organonitrogen compound
- Organic oxygen compound
- Organooxygen compound
- Organic nitrogen compound
- Carbonyl group
- Hydrocarbon derivative
- Organopnictogen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0092-8549000000-e95459cdd2a46f8d9827 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a6r-0109700000-42cd526479b05f760baf | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-004i-0009000000-6be65d5ffdb842c39ae9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-01bj-9452000000-b3f885328291dff5a80f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-056r-0009300000-e0e5e86cbd130b0b112e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-01r5-3329100000-7b31bc961f78ffff57f5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0006-9203000000-b6c6c4b5a7a7649f3dd7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0006-0098300000-1ac127cc4a33497a1000 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-014i-1189000000-d4e73b46a3c7ebe78d27 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-014i-6896000000-d7c1cf0b191abdd8d192 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0uxu-5429100000-681578a6e3cea0de9fff | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00kf-2089000000-a2419c1466bc3b8f9137 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0006-6195000000-1d178466438140b00525 | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0039126 |
|---|
| FooDB ID | FDB018638 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 167683 |
|---|
| ChEBI ID | 165847 |
|---|
| PubChem Compound ID | 193232 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|