| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 03:20:30 UTC |
|---|
| Update Date | 2016-11-09 01:19:27 UTC |
|---|
| Accession Number | CHEM032372 |
|---|
| Identification |
|---|
| Common Name | Necatorine |
|---|
| Class | Small Molecule |
|---|
| Description | Necatorine is found in mushrooms. Necatorine is an alkaloid from the mushroom Lactarius necator. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
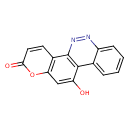
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 5-Hydroxycoumaro(7,8-c)cinnoline | MeSH | | 5-Hydroxycoumaro(6,7-c)cinnoline | MeSH | | 11-Hydroxy-2H-benzo(c)pyrano(2,3-H)cinnolin-2-one | HMDB | | 11-Hydroxy-2H-benzo[c]pyrano[2,3-H]cinnolin-2-one, 9ci | HMDB | | Necatorin | HMDB | | Carbon tetrachloride | ChEBI | | CCL4 | ChEBI | | Kohlenstofftetrachlorid | ChEBI | | Tetra | ChEBI | | Tetrachloridocarbon | ChEBI | | Tetrachlorkohlenstoff | ChEBI | | Tetrachlormethan | ChEBI | | Tetrachloromethane | KEGG | | Carbon chloride? | HMDB | | Carbon tet | HMDB | | Carbontetrachloride | HMDB | | F10 | HMDB | | Freon 10 | HMDB | | Halon 104 | HMDB | | Halon 1040 | HMDB | | HSDB 53 | HMDB | | Methane tetrachloride | HMDB | | Perchloromethane | HMDB | | R10 | HMDB | | Refrigerant R10 | HMDB | | Tetrachloro-methane | HMDB | | Tetrachloromethane, 9ci | HMDB | | Tetrasol | HMDB | | Thawpit | HMDB | | Univerm | HMDB | | Vermoestricid | HMDB | | Tetrachloride, carbon | HMDB |
|
|---|
| Chemical Formula | C15H8N2O3 |
|---|
| Average Molecular Mass | 264.236 g/mol |
|---|
| Monoisotopic Mass | 264.053 g/mol |
|---|
| CAS Registry Number | 89915-35-5 |
|---|
| IUPAC Name | 9-hydroxy-6-oxa-17,18-diazatetracyclo[8.8.0.0²,⁷.0¹¹,¹⁶]octadeca-1(10),2(7),3,8,11,13,15,17-octaen-5-one |
|---|
| Traditional Name | 9-hydroxy-6-oxa-17,18-diazatetracyclo[8.8.0.0²,⁷.0¹¹,¹⁶]octadeca-1(10),2(7),3,8,11,13,15,17-octaen-5-one |
|---|
| SMILES | OC1=CC2=C(C=CC(=O)O2)C2=C1C1=CC=CC=C1N=N2 |
|---|
| InChI Identifier | InChI=1S/C15H8N2O3/c18-11-7-12-9(5-6-13(19)20-12)15-14(11)8-3-1-2-4-10(8)16-17-15/h1-7,18H |
|---|
| InChI Key | WNQBVKOXDIYRFH-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as coumarins and derivatives. These are polycyclic aromatic compounds containing a 1-benzopyran moiety with a ketone group at the C2 carbon atom (1-benzopyran-2-one). |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Phenylpropanoids and polyketides |
|---|
| Class | Coumarins and derivatives |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Coumarins and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Coumarin
- Diazanaphthalene
- Benzopyran
- Cinnoline
- 1-benzopyran
- 1-hydroxy-2-unsubstituted benzenoid
- Pyranone
- Benzenoid
- Pyridazine
- Pyran
- Heteroaromatic compound
- Lactone
- Oxacycle
- Azacycle
- Organoheterocyclic compound
- Organic nitrogen compound
- Hydrocarbon derivative
- Organooxygen compound
- Organonitrogen compound
- Organic oxide
- Organopnictogen compound
- Organic oxygen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-029i-0090000000-3c5132bac5f8ee787484 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (1 TMS) - 70eV, Positive | splash10-00di-9086000000-be8ab3c5811db476eff7 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014i-0090000000-18a1ba1992601177dfc2 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-014i-0090000000-2bc1d8302ed38aa8a2f1 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-00kb-1490000000-a18f54f87e342204f47d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03di-0090000000-a24d2e3f3e4b0d59fc65 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-03di-0090000000-e9ff52348ee37b35e571 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-014l-0690000000-ee636bfabc1235026f0c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03di-0090000000-3fa7cc4700cbf2f24722 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-03di-0090000000-3fa7cc4700cbf2f24722 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-014i-0090000000-a65e42cbc7521c1de576 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014i-0090000000-6315ae722cd8f52a48b9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-014i-0090000000-6315ae722cd8f52a48b9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-00ri-0090000000-fdfafe7fa5a399554c51 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0039035 |
|---|
| FooDB ID | FDB018532 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Carbon_Tetrachloride |
|---|
| Chemspider ID | 5730 |
|---|
| ChEBI ID | 27385 |
|---|
| PubChem Compound ID | 135554414 |
|---|
| Kegg Compound ID | C07561 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|