| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 03:20:17 UTC |
|---|
| Update Date | 2016-11-09 01:19:27 UTC |
|---|
| Accession Number | CHEM032366 |
|---|
| Identification |
|---|
| Common Name | Pyranomammea B |
|---|
| Class | Small Molecule |
|---|
| Description | Pyranomammea B is found in fruits. Pyranomammea B is a constituent of Mammea americana (mamey). |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
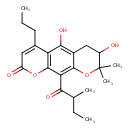
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Mammea b/bb cyclo e | HMDB |
|
|---|
| Chemical Formula | C22H28O6 |
|---|
| Average Molecular Mass | 388.454 g/mol |
|---|
| Monoisotopic Mass | 388.189 g/mol |
|---|
| CAS Registry Number | Not Available |
|---|
| IUPAC Name | 5,7-dihydroxy-8,8-dimethyl-10-(2-methylbutanoyl)-4-propyl-2H,6H,7H,8H-pyrano[3,2-g]chromen-2-one |
|---|
| Traditional Name | 5,7-dihydroxy-8,8-dimethyl-10-(2-methylbutanoyl)-4-propyl-6H,7H-pyrano[3,2-g]chromen-2-one |
|---|
| SMILES | CCCC1=CC(=O)OC2=C1C(O)=C1CC(O)C(C)(C)OC1=C2C(=O)C(C)CC |
|---|
| InChI Identifier | InChI=1S/C22H28O6/c1-6-8-12-9-15(24)27-21-16(12)19(26)13-10-14(23)22(4,5)28-20(13)17(21)18(25)11(3)7-2/h9,11,14,23,26H,6-8,10H2,1-5H3 |
|---|
| InChI Key | IWAUBOJXTGEZNN-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as linear pyranocoumarins. These are organic compounds containing a pyran (or a hydrogenated derivative) linearly fused to a coumarin moiety. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Phenylpropanoids and polyketides |
|---|
| Class | Coumarins and derivatives |
|---|
| Sub Class | Pyranocoumarins |
|---|
| Direct Parent | Linear pyranocoumarins |
|---|
| Alternative Parents | |
|---|
| Substituents | - Linear pyranocoumarin
- Pyranochromene
- 2,2-dimethyl-1-benzopyran
- Butyrophenone
- Chromane
- Benzopyran
- 1-benzopyran
- Aryl alkyl ketone
- Aryl ketone
- Alkyl aryl ether
- Pyranone
- Benzenoid
- Pyran
- Heteroaromatic compound
- Ketone
- Lactone
- Secondary alcohol
- Organoheterocyclic compound
- Ether
- Oxacycle
- Alcohol
- Hydrocarbon derivative
- Organic oxide
- Organic oxygen compound
- Organooxygen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0ab9-1009000000-2d0a4efc67d590d72508 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (2 TMS) - 70eV, Positive | splash10-014i-8110390000-4cfc0190bb3cada7a126 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-000i-1019000000-7facc5baec22608c34f2 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0670-3029000000-0f8bea97887355324197 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0ap3-9050000000-038b29e5b13ca74763b3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-000i-0009000000-443a15e5e512689978ca | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0079-9047000000-27e62dd22f8bec5b3997 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-014r-6091000000-c9a4a345df16041a9dfc | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-000i-0009000000-1bd462d2f1db9df6b735 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-000i-0009000000-432f930cd05d7db8daa3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-00kr-9035000000-6f5cab4a5f5b99de8829 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-000i-0009000000-7f16ac45973a6479b3e5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-000i-0009000000-4029e9517e60896d8cca | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0f7a-0196000000-d67a7d9b6b6cc554ab90 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0039029 |
|---|
| FooDB ID | FDB018525 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 35014723 |
|---|
| ChEBI ID | 175950 |
|---|
| PubChem Compound ID | 131752522 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|