| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 03:15:23 UTC |
|---|
| Update Date | 2016-11-09 01:19:26 UTC |
|---|
| Accession Number | CHEM032256 |
|---|
| Identification |
|---|
| Common Name | Armillarivin |
|---|
| Class | Small Molecule |
|---|
| Description | Armillarivin is found in mushrooms. Armillarivin is a metabolite of Armillaria mellea (honey mushroom). |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
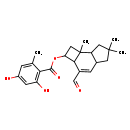
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 3-Formyl-6,6,7b-trimethyl-1H,2H,2ah,4ah,5H,6H,7H,7ah,7BH-cyclobuta[e]inden-2-yl 2,4-dihydroxy-6-methylbenzoic acid | HMDB |
|
|---|
| Chemical Formula | C23H28O5 |
|---|
| Average Molecular Mass | 384.465 g/mol |
|---|
| Monoisotopic Mass | 384.194 g/mol |
|---|
| CAS Registry Number | 135247-97-1 |
|---|
| IUPAC Name | 3-formyl-6,6,7b-trimethyl-1H,2H,2aH,4aH,5H,6H,7H,7aH,7bH-cyclobuta[e]inden-2-yl 2,4-dihydroxy-6-methylbenzoate |
|---|
| Traditional Name | 3-formyl-6,6,7b-trimethyl-1H,2H,2aH,4aH,5H,7H,7aH-cyclobuta[e]inden-2-yl 2,4-dihydroxy-6-methylbenzoate |
|---|
| SMILES | CC1=CC(O)=CC(O)=C1C(=O)OC1CC2(C)C1C(C=O)=CC1CC(C)(C)CC21 |
|---|
| InChI Identifier | InChI=1S/C23H28O5/c1-12-5-15(25)7-17(26)19(12)21(27)28-18-10-23(4)16-9-22(2,3)8-13(16)6-14(11-24)20(18)23/h5-7,11,13,16,18,20,25-26H,8-10H2,1-4H3 |
|---|
| InChI Key | NFCOJBIECSQMAM-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as melleolides and analogues. Melleolides and analogues are compounds with a structure characterized by the presence of a 2-hydroxy-4-methoxy-6-methylbenzoic acid derivative linked to a 3,6,6,7b-tetramethyl-cyclobuta[e]indene moiety. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Prenol lipids |
|---|
| Sub Class | Sesquiterpenoids |
|---|
| Direct Parent | Melleolides and analogues |
|---|
| Alternative Parents | |
|---|
| Substituents | - Melleolide-skeleton
- P-hydroxybenzoic acid alkyl ester
- P-hydroxybenzoic acid ester
- O-hydroxybenzoic acid ester
- Dihydroxybenzoic acid
- Benzoate ester
- Salicylic acid or derivatives
- Benzoic acid or derivatives
- M-cresol
- Resorcinol
- Benzoyl
- 1-hydroxy-4-unsubstituted benzenoid
- 1-hydroxy-2-unsubstituted benzenoid
- Toluene
- Phenol
- Monocyclic benzene moiety
- Benzenoid
- Vinylogous acid
- Carboxylic acid ester
- Carboxylic acid derivative
- Monocarboxylic acid or derivatives
- Carbonyl group
- Organooxygen compound
- Hydrocarbon derivative
- Organic oxide
- Organic oxygen compound
- Aldehyde
- Aromatic homopolycyclic compound
|
|---|
| Molecular Framework | Aromatic homopolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0f7c-1921000000-599cec08d570dc0ad927 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (2 TMS) - 70eV, Positive | splash10-000l-1950110000-4865aef9adfb6f1b17ec | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-000i-0439000000-a09231d868b877a61d06 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0uxr-0953000000-89ef4a2067604adb080f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0udi-2910000000-dae424cc6cf5bda32a36 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-001i-0229000000-b209c00ac580055c1089 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00si-0926000000-d2ea6d0fc734d18ba59c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-00di-2920000000-ec363da0b7c1951b6480 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-000i-0869000000-f7314a5e31de0522e528 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0gc9-2945000000-00e2180a120f2a6bd6c6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0pbi-1920000000-edae8435a4206adc198a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-01c0-0904000000-00f04b36257385d08a35 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00di-2921000000-291dd6ad9075b3012787 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-00dl-8931000000-14fb3f5e1641dd26d857 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0038917 |
|---|
| FooDB ID | FDB018381 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00055512 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 35014691 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 78171719 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|