| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 03:09:49 UTC |
|---|
| Update Date | 2016-11-09 01:19:24 UTC |
|---|
| Accession Number | CHEM032130 |
|---|
| Identification |
|---|
| Common Name | Melleolide E |
|---|
| Class | Small Molecule |
|---|
| Description | Melleolide E is found in mushrooms. Melleolide E is a metabolite of Armillaria mellea (honey mushroom). |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
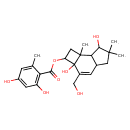
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 2a,7-Dihydroxy-3-(hydroxymethyl)-6,6,7b-trimethyl-1H,2H,2ah,4ah,5H,6H,7H,7ah,7BH-cyclobuta[e]inden-2-yl 2,4-dihydroxy-6-methylbenzoic acid | HMDB |
|
|---|
| Chemical Formula | C23H30O7 |
|---|
| Average Molecular Mass | 418.480 g/mol |
|---|
| Monoisotopic Mass | 418.199 g/mol |
|---|
| CAS Registry Number | 117258-73-8 |
|---|
| IUPAC Name | 2a,7-dihydroxy-3-(hydroxymethyl)-6,6,7b-trimethyl-1H,2H,2aH,4aH,5H,6H,7H,7aH,7bH-cyclobuta[e]inden-2-yl 2,4-dihydroxy-6-methylbenzoate |
|---|
| Traditional Name | 2a,7-dihydroxy-3-(hydroxymethyl)-6,6,7b-trimethyl-1H,2H,4aH,5H,7H,7aH-cyclobuta[e]inden-2-yl 2,4-dihydroxy-6-methylbenzoate |
|---|
| SMILES | CC1=CC(O)=CC(O)=C1C(=O)OC1CC2(C)C3C(CC(C)(C)C3O)C=C(CO)C12O |
|---|
| InChI Identifier | InChI=1S/C23H30O7/c1-11-5-14(25)7-15(26)17(11)20(28)30-16-9-22(4)18-12(8-21(2,3)19(18)27)6-13(10-24)23(16,22)29/h5-7,12,16,18-19,24-27,29H,8-10H2,1-4H3 |
|---|
| InChI Key | SWVWDNGFQHDELB-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as melleolides and analogues. Melleolides and analogues are compounds with a structure characterized by the presence of a 2-hydroxy-4-methoxy-6-methylbenzoic acid derivative linked to a 3,6,6,7b-tetramethyl-cyclobuta[e]indene moiety. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Prenol lipids |
|---|
| Sub Class | Sesquiterpenoids |
|---|
| Direct Parent | Melleolides and analogues |
|---|
| Alternative Parents | |
|---|
| Substituents | - Melleolide-skeleton
- P-hydroxybenzoic acid alkyl ester
- P-hydroxybenzoic acid ester
- O-hydroxybenzoic acid ester
- Dihydroxybenzoic acid
- Benzoate ester
- Salicylic acid or derivatives
- Benzoic acid or derivatives
- M-cresol
- Benzoyl
- Resorcinol
- 1-hydroxy-4-unsubstituted benzenoid
- 1-hydroxy-2-unsubstituted benzenoid
- Phenol
- Toluene
- Benzenoid
- Monocyclic benzene moiety
- Vinylogous acid
- Cyclic alcohol
- Tertiary alcohol
- Carboxylic acid ester
- Cyclobutanol
- Secondary alcohol
- Carboxylic acid derivative
- Monocarboxylic acid or derivatives
- Alcohol
- Organooxygen compound
- Primary alcohol
- Hydrocarbon derivative
- Organic oxide
- Organic oxygen compound
- Aromatic homopolycyclic compound
|
|---|
| Molecular Framework | Aromatic homopolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0kmj-5970000000-2902c480cc475e64e86b | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (3 TMS) - 70eV, Positive | splash10-0002-0091000000-680c1a0d79765fc04f9e | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0udi-0232900000-17a65b176359e195602f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0ue9-0945400000-0853505451596b673924 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0ue9-2940000000-9c00080c14c17ec28d2c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-014i-0515900000-681616a91628af2ac4c4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-01ba-0958200000-4e1780b1b0cc7622b5f2 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-00di-1930000000-fc74e5ba8fcf80c7b527 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-014i-0304900000-af057929185280a1ed06 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00di-1941200000-b380fb6e7ab9a885a75f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-00xu-9721000000-f5780d29accb7314a4c1 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0uyi-0582900000-17e2b9eac4d884e83ade | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0ue9-4941300000-5bb19a1bca972c93afc1 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0uei-5912000000-42bf78ba5f717def401e | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0038785 |
|---|
| FooDB ID | FDB018207 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00056547 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 35014659 |
|---|
| ChEBI ID | 175349 |
|---|
| PubChem Compound ID | 14166116 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|