| Identification |
|---|
| Common Name | Gibberellin A4 glucosyl ester |
|---|
| Class | Small Molecule |
|---|
| Description | Not Available |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
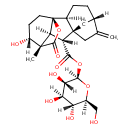
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (2R,3S,4R,5R,6S)-3,4,5-Trihydroxy-6-(hydroxymethyl)oxan-2-yl (1S,2S,5S,8S,9R,10S,11S,12R)-12-hydroxy-11-methyl-6-methylidene-16-oxo-15-oxapentacyclo[9.3.2.1,.0,.0,]heptadecane-9-carboxylic acid | Generator | | (2R,3S,4R,5R,6S)-3,4,5-Trihydroxy-6-(hydroxymethyl)oxan-2-yl (1S,2S,5S,8S,9R,10S,11S,12R)-12-hydroxy-11-methyl-6-methylidene-16-oxo-15-oxapentacyclo[9.3.2.1⁵,⁸.0¹,¹⁰.0²,⁸]heptadecane-9-carboxylic acid | Generator |
|
|---|
| Chemical Formula | C25H34O10 |
|---|
| Average Molecular Mass | 494.537 g/mol |
|---|
| Monoisotopic Mass | 494.215 g/mol |
|---|
| CAS Registry Number | 54788-52-2 |
|---|
| IUPAC Name | (2R,3S,4R,5R,6S)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl (1S,2S,5S,8S,9R,10S,11S,12R)-12-hydroxy-11-methyl-6-methylidene-16-oxo-15-oxapentacyclo[9.3.2.1⁵,⁸.0¹,¹⁰.0²,⁸]heptadecane-9-carboxylate |
|---|
| Traditional Name | (2R,3S,4R,5R,6S)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl (1S,2S,5S,8S,9R,10S,11S,12R)-12-hydroxy-11-methyl-6-methylidene-16-oxo-15-oxapentacyclo[9.3.2.1⁵,⁸.0¹,¹⁰.0²,⁸]heptadecane-9-carboxylate |
|---|
| SMILES | [H][C@]12C[C@]3(CC1=C)[C@]([H])(CC2)[C@]12CC[C@@]([H])(O)[C@@](C)(C(=O)O1)[C@]2([H])[C@@]3([H])C(=O)O[C@@]1([H])O[C@@]([H])(CO)[C@]([H])(O)[C@@]([H])(O)[C@]1([H])O |
|---|
| InChI Identifier | InChI=1S/C25H34O10/c1-10-7-24-8-11(10)3-4-13(24)25-6-5-14(27)23(2,22(32)35-25)19(25)15(24)20(31)34-21-18(30)17(29)16(28)12(9-26)33-21/h11-19,21,26-30H,1,3-9H2,2H3/t11-,12-,13-,14+,15-,16-,17+,18-,19-,21+,23+,24+,25-/m0/s1 |
|---|
| InChI Key | WAJNZXFSVANOAB-WBLXFBQTSA-N |
|---|