| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 02:57:38 UTC |
|---|
| Update Date | 2016-11-09 01:19:20 UTC |
|---|
| Accession Number | CHEM031838 |
|---|
| Identification |
|---|
| Common Name | Auxin a |
|---|
| Class | Small Molecule |
|---|
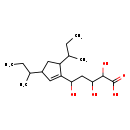
| Description | A carbocyclic compound that is 3,5-di-sec-butylcyclopentene which is substituted by a 4-carboxy-1,3,4-trihydroxybutyl group at position 1. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 5-(2,4-Di-sec-butyl-cyclopent-5-enyl)-2,3,5-trihydroxy-valeric acid | ChEBI | | 5-(3,5-Di-sec-butylcyclopent-1-enyl)-2,3,5-trihydroxyvaleric acid | ChEBI | | 5-C-(3,5-Bis(1-methylpropyl)-1-cyclopenten-1-yl)-4-deoxy-pentonic acid | ChEBI | | Auxen triolic acid | ChEBI | | Auxentriolic acid | ChEBI | | Auxin-a | ChEBI | | 5-(2,4-Di-sec-butyl-cyclopent-5-enyl)-2,3,5-trihydroxy-valerate | Generator | | 5-(3,5-Di-sec-butylcyclopent-1-enyl)-2,3,5-trihydroxyvalerate | Generator | | 5-C-(3,5-Bis(1-methylpropyl)-1-cyclopenten-1-yl)-4-deoxy-pentonate | Generator | | Auxen triolate | Generator | | Auxentriolate | Generator | | Acids, indolylacetic | HMDB | | Auxin | HMDB | | Indoleacetic acids | HMDB | | Auxins | HMDB | | Acids, indoleacetic | HMDB | | Indolylacetic acids | HMDB | | 5-[3,5-Bis(butan-2-yl)cyclopent-1-en-1-yl]-2,3,5-trihydroxypentanoate | HMDB |
|
|---|
| Chemical Formula | C18H32O5 |
|---|
| Average Molecular Mass | 328.444 g/mol |
|---|
| Monoisotopic Mass | 328.225 g/mol |
|---|
| CAS Registry Number | Not Available |
|---|
| IUPAC Name | 5-[3,5-bis(butan-2-yl)cyclopent-1-en-1-yl]-2,3,5-trihydroxypentanoic acid |
|---|
| Traditional Name | 5-[3,5-bis(sec-butyl)cyclopent-1-en-1-yl]-2,3,5-trihydroxypentanoic acid |
|---|
| SMILES | CCC(C)C1CC(C(C)CC)C(=C1)C(O)CC(O)C(O)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C18H32O5/c1-5-10(3)12-7-13(11(4)6-2)14(8-12)15(19)9-16(20)17(21)18(22)23/h8,10-13,15-17,19-21H,5-7,9H2,1-4H3,(H,22,23) |
|---|
| InChI Key | RKOUCPMGBBKLNK-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as monocyclic monoterpenoids. These are monoterpenoids containing 1 ring in the isoprene chain. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Prenol lipids |
|---|
| Sub Class | Monoterpenoids |
|---|
| Direct Parent | Monocyclic monoterpenoids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Monocyclic monoterpenoid
- Medium-chain hydroxy acid
- Medium-chain fatty acid
- Beta-hydroxy acid
- Hydroxy fatty acid
- Alpha-hydroxy acid
- Hydroxy acid
- Fatty acyl
- Monosaccharide
- Secondary alcohol
- Carboxylic acid derivative
- Carboxylic acid
- Polyol
- Monocarboxylic acid or derivatives
- Alcohol
- Carbonyl group
- Hydrocarbon derivative
- Organic oxygen compound
- Organooxygen compound
- Organic oxide
- Aliphatic homomonocyclic compound
|
|---|
| Molecular Framework | Aliphatic homomonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-03fs-8694000000-2d35c05df67e8b959530 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (4 TMS) - 70eV, Positive | splash10-0zfr-5022197000-aea91607506891fc1dc2 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-03fu-0198000000-96abea4dfa87529fb828 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a4l-1291000000-fa556f72c2005293532f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-014i-9430000000-e6d21fcf98ed47739931 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-4297000000-986195f9ca9e0e895996 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0ayi-4290000000-fae70c80e633627d3c1f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-05i0-9150000000-503ad70891bf9201b4e0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-056r-0019000000-95245e0314849ad29cbb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-056r-9171000000-d6ad5da9d6fe3112435b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-00di-2930000000-6a7301ca3bba8060a7a8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-004i-0289000000-0a473c8121ab47402c74 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00fr-0910000000-eaf6d600df373ecb4034 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a4i-9210000000-edf209bbae72dc344554 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0038483 |
|---|
| FooDB ID | FDB017851 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00001524 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 83749 |
|---|
| ChEBI ID | 142254 |
|---|
| PubChem Compound ID | 92772 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|