| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 02:55:53 UTC |
|---|
| Update Date | 2016-11-09 01:19:20 UTC |
|---|
| Accession Number | CHEM031802 |
|---|
| Identification |
|---|
| Common Name | 5,7-Dimethoxy-6-methylflavanone |
|---|
| Class | Small Molecule |
|---|
| Description | 5,7-Dimethoxy-6-methylflavanone is found in tea. 5,7-Dimethoxy-6-methylflavanone is a constituent of Leptospermum scoparium (red tea). |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
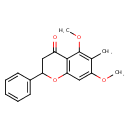
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 5,7-Dimethoxy-6-C-methylflavanone | HMDB |
|
|---|
| Chemical Formula | C18H18O4 |
|---|
| Average Molecular Mass | 298.333 g/mol |
|---|
| Monoisotopic Mass | 298.121 g/mol |
|---|
| CAS Registry Number | 128508-16-7 |
|---|
| IUPAC Name | 5,7-dimethoxy-6-methyl-2-phenyl-3,4-dihydro-2H-1-benzopyran-4-one |
|---|
| Traditional Name | 5,7-dimethoxy-6-methyl-2-phenyl-2,3-dihydro-1-benzopyran-4-one |
|---|
| SMILES | COC1=CC2=C(C(=O)CC(O2)C2=CC=CC=C2)C(OC)=C1C |
|---|
| InChI Identifier | InChI=1S/C18H18O4/c1-11-14(20-2)10-16-17(18(11)21-3)13(19)9-15(22-16)12-7-5-4-6-8-12/h4-8,10,15H,9H2,1-3H3 |
|---|
| InChI Key | XYMPWWTYELQXGU-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as 7-o-methylated flavonoids. These are flavonoids with methoxy groups attached to the C7 atom of the flavonoid backbone. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Phenylpropanoids and polyketides |
|---|
| Class | Flavonoids |
|---|
| Sub Class | O-methylated flavonoids |
|---|
| Direct Parent | 7-O-methylated flavonoids |
|---|
| Alternative Parents | |
|---|
| Substituents | - 5-methoxyflavonoid-skeleton
- 7-methoxyflavonoid-skeleton
- Flavanone
- Flavan
- Chromone
- Chromane
- Benzopyran
- 1-benzopyran
- Anisole
- Aryl alkyl ketone
- Aryl ketone
- Alkyl aryl ether
- Monocyclic benzene moiety
- Benzenoid
- Ketone
- Ether
- Organoheterocyclic compound
- Oxacycle
- Organic oxide
- Hydrocarbon derivative
- Organic oxygen compound
- Organooxygen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-00lr-1290000000-38646b7b74645a022ecd | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0002-0290000000-a62ee2ddff938fe77c92 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0002-2690000000-494b5097e9e1db4d9134 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0ap0-2900000000-49fdc3f2586e74f20878 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0002-0190000000-50dd932bd56556ec56f3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0002-1390000000-e2e46cdc78bce454ad2d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0ug0-5940000000-2f2d5a54aaf629575a07 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0002-0490000000-e6e2f171ed3c8a20587f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0002-0900000000-4141617a39b42143a025 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0f6t-0900000000-b36aeb2f66c56162d1b6 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0038453 |
|---|
| FooDB ID | FDB017812 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00008420 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 22370032 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 14583632 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|