| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 02:54:02 UTC |
|---|
| Update Date | 2016-11-09 01:19:19 UTC |
|---|
| Accession Number | CHEM031760 |
|---|
| Identification |
|---|
| Common Name | Glucohesperalin |
|---|
| Class | Small Molecule |
|---|
| Description | Glucohesperalin is found in fats and oils. Glucohesperalin is present in seeds of Hesperis matronalis (sweet rocket). |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
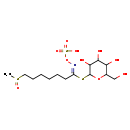
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 6-(Methylsulfinyl)hexyl glucosinolate | HMDB | | 6-Methylsulfinylhexyl glucosinolate | HMDB | | Glucohesperin | HMDB | | {[(e)-(7-methanesulfinyl-1-{[3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]sulfanyl}heptylidene)amino]oxy}sulfonate | Generator | | {[(e)-(7-methanesulphinyl-1-{[3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]sulphanyl}heptylidene)amino]oxy}sulphonate | Generator | | {[(e)-(7-methanesulphinyl-1-{[3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]sulphanyl}heptylidene)amino]oxy}sulphonic acid | Generator |
|
|---|
| Chemical Formula | C14H27NO10S3 |
|---|
| Average Molecular Mass | 465.560 g/mol |
|---|
| Monoisotopic Mass | 465.080 g/mol |
|---|
| CAS Registry Number | 33049-17-1 |
|---|
| IUPAC Name | {[(E)-(7-methanesulfinyl-1-{[3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]sulfanyl}heptylidene)amino]oxy}sulfonic acid |
|---|
| Traditional Name | [(E)-(7-methanesulfinyl-1-{[3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]sulfanyl}heptylidene)amino]oxysulfonic acid |
|---|
| SMILES | CS(=O)CCCCCC\C(SC1OC(CO)C(O)C(O)C1O)=N/OS(O)(=O)=O |
|---|
| InChI Identifier | InChI=1S/C14H27NO10S3/c1-27(20)7-5-3-2-4-6-10(15-25-28(21,22)23)26-14-13(19)12(18)11(17)9(8-16)24-14/h9,11-14,16-19H,2-8H2,1H3,(H,21,22,23)/b15-10+ |
|---|
| InChI Key | OOGAQHVYHLPICD-XNTDXEJSSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as alkylglucosinolates. These are organic compounds containing a glucosinolate moiety that carries an alkyl chain. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic oxygen compounds |
|---|
| Class | Organooxygen compounds |
|---|
| Sub Class | Carbohydrates and carbohydrate conjugates |
|---|
| Direct Parent | Alkylglucosinolates |
|---|
| Alternative Parents | |
|---|
| Substituents | - Alkylglucosinolate
- Glycosyl compound
- S-glycosyl compound
- Oxane
- Monothioacetal
- Organic sulfuric acid or derivatives
- Secondary alcohol
- Sulfoxide
- Sulfinyl compound
- Oxacycle
- Polyol
- Sulfenyl compound
- Organoheterocyclic compound
- Alcohol
- Primary alcohol
- Hydrocarbon derivative
- Organosulfur compound
- Organonitrogen compound
- Organic oxide
- Organopnictogen compound
- Organic nitrogen compound
- Aliphatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aliphatic heteromonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0udj-9702600000-0a7bd66ccff2a6fdfd49 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (3 TMS) - 70eV, Positive | splash10-014i-5584219000-e5d01896bdbccccb9620 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0002-0814900000-a787a820d9c344b09fe1 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-114r-0849000000-cfed2c8f64a9f3598933 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a4v-9320000000-f7984fdc55b1b9cbb218 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0ik9-9125100000-23d13447e9008804c079 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-03di-9100000000-561fdf7a0251d592b372 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-03di-9510000000-7f9c955b2f0cb4b7f8d8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014i-0000900000-9bec2ca8b1a60790af05 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0udi-2026900000-fe1308b694217f581a4a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0hi6-2079000000-37933342635a2ea626d0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03di-5000900000-49ffb759be68b70ad9d4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0w2i-9128700000-7e2fc453da6ddc83f986 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-01px-9340000000-5c5553c8095d68c8bef1 | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0038410 |
|---|
| FooDB ID | FDB017764 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00007348 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 24808181 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 14574455 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|