| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 02:36:31 UTC |
|---|
| Update Date | 2016-11-09 01:19:15 UTC |
|---|
| Accession Number | CHEM031379 |
|---|
| Identification |
|---|
| Common Name | Quercetin 3-rutinoside 4'-glucoside |
|---|
| Class | Small Molecule |
|---|
| Description | Not Available |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
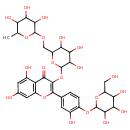
| Chemical Structure | |
|---|
| Synonyms | Not Available |
|---|
| Chemical Formula | C33H40O21 |
|---|
| Average Molecular Mass | 772.658 g/mol |
|---|
| Monoisotopic Mass | 772.206 g/mol |
|---|
| CAS Registry Number | 89439-59-8 |
|---|
| IUPAC Name | 5,7-dihydroxy-2-(3-hydroxy-4-{[3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}phenyl)-3-[(3,4,5-trihydroxy-6-{[(3,4,5-trihydroxy-6-methyloxan-2-yl)oxy]methyl}oxan-2-yl)oxy]-4H-chromen-4-one |
|---|
| Traditional Name | 5,7-dihydroxy-2-(3-hydroxy-4-{[3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}phenyl)-3-[(3,4,5-trihydroxy-6-{[(3,4,5-trihydroxy-6-methyloxan-2-yl)oxy]methyl}oxan-2-yl)oxy]chromen-4-one |
|---|
| SMILES | CC1OC(OCC2OC(OC3=C(OC4=C(C(O)=CC(O)=C4)C3=O)C3=CC(O)=C(OC4OC(CO)C(O)C(O)C4O)C=C3)C(O)C(O)C2O)C(O)C(O)C1O |
|---|
| InChI Identifier | InChI=1S/C33H40O21/c1-9-19(38)23(42)26(45)31(49-9)48-8-17-21(40)25(44)28(47)33(53-17)54-30-22(41)18-13(37)5-11(35)6-15(18)50-29(30)10-2-3-14(12(36)4-10)51-32-27(46)24(43)20(39)16(7-34)52-32/h2-6,9,16-17,19-21,23-28,31-40,42-47H,7-8H2,1H3 |
|---|
| InChI Key | VLWGTVNAOQPEJO-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as flavonoid-3-o-glycosides. These are phenolic compounds containing a flavonoid moiety which is O-glycosidically linked to carbohydrate moiety at the C3-position. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Phenylpropanoids and polyketides |
|---|
| Class | Flavonoids |
|---|
| Sub Class | Flavonoid glycosides |
|---|
| Direct Parent | Flavonoid-3-O-glycosides |
|---|
| Alternative Parents | |
|---|
| Substituents | - Flavonoid-3-o-glycoside
- Hydroxyflavonoid
- Flavone
- 7-hydroxyflavonoid
- 5-hydroxyflavonoid
- 3'-hydroxyflavonoid
- Phenolic glycoside
- O-glycosyl compound
- Glycosyl compound
- Chromone
- Disaccharide
- Benzopyran
- 1-benzopyran
- Phenoxy compound
- Phenol ether
- 1-hydroxy-4-unsubstituted benzenoid
- 1-hydroxy-2-unsubstituted benzenoid
- Phenol
- Pyranone
- Monocyclic benzene moiety
- Oxane
- Benzenoid
- Pyran
- Heteroaromatic compound
- Vinylogous acid
- Secondary alcohol
- Acetal
- Oxacycle
- Organoheterocyclic compound
- Polyol
- Organic oxide
- Alcohol
- Hydrocarbon derivative
- Primary alcohol
- Organic oxygen compound
- Organooxygen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0aov-0213954800-18a5f46bffe71a566176 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0udi-0309822100-bb8b0e36faad677f621d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0udi-1629311100-c160fe446fe379f17df2 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0mb9-4713415900-5386165859a2bd7f53bd | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-03dj-4934823300-a5d827194c32774bb8d9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-03dm-5669401000-bb6f80dedee93416f512 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014i-0000900200-6490cc53d75b11ebcf81 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-01i0-0000900900-19191d74d2ebf6d20e1e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-014i-0000900000-ab6c9730050acb7a41bb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-00di-0000000900-0dd3d7eb0a0579e613c9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0229-0000500900-93d5412fed5fdb659bc7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-03di-0010900100-f7f0f763693fffc2a1a2 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0303634 |
|---|
| FooDB ID | FDB017130 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | Not Available |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|