| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 02:33:03 UTC |
|---|
| Update Date | 2016-11-09 01:19:14 UTC |
|---|
| Accession Number | CHEM031301 |
|---|
| Identification |
|---|
| Common Name | 4-Butyl-2,5-dimethyloxazole |
|---|
| Class | Small Molecule |
|---|
| Description | 4-Butyl-2,5-dimethyloxazole is found in animal foods. 4-Butyl-2,5-dimethyloxazole is a constituent of roast beef, cocoa butter and French fried potato aroma. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
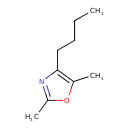
| Chemical Structure | |
|---|
| Synonyms | Not Available |
|---|
| Chemical Formula | C9H15NO |
|---|
| Average Molecular Mass | 153.222 g/mol |
|---|
| Monoisotopic Mass | 153.115 g/mol |
|---|
| CAS Registry Number | 30408-62-9 |
|---|
| IUPAC Name | 4-butyl-2,5-dimethyl-1,3-oxazole |
|---|
| Traditional Name | 4-butyl-2,5-dimethyl-1,3-oxazole |
|---|
| SMILES | CCCCC1=C(C)OC(C)=N1 |
|---|
| InChI Identifier | InChI=1S/C9H15NO/c1-4-5-6-9-7(2)11-8(3)10-9/h4-6H2,1-3H3 |
|---|
| InChI Key | MRXQRKKZDKCDSE-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as 2,4,5-trisubstituted oxazoles. 2,4,5-trisubstituted oxazoles are compounds containing an oxazole ring substituted at positions 2, 4 and 5 only. Oxazole is a five-membered aromatic heterocycle with one oxygen, one nitrogen, and three carbon atoms. Isomers include 1,2-oxazole and 1,3-oxazole. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Azoles |
|---|
| Sub Class | Oxazoles |
|---|
| Direct Parent | 2,4,5-trisubstituted oxazoles |
|---|
| Alternative Parents | |
|---|
| Substituents | - 2,4,5-trisubstituted 1,3-oxazole
- Heteroaromatic compound
- Oxacycle
- Azacycle
- Organic nitrogen compound
- Organic oxygen compound
- Organopnictogen compound
- Hydrocarbon derivative
- Organooxygen compound
- Organonitrogen compound
- Aromatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aromatic heteromonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-01tc-9700000000-f40e776eef15d6c559f6 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0udi-0900000000-929abc2500a3bb2e13aa | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0udi-7900000000-19013a1e68e954dc5e3b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0f6x-9000000000-4aea967ba778a21df7cb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0zfr-0900000000-1e865dacff9c3f39c0a9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0nn9-3900000000-2dd9255105d70580b8db | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0f7o-9000000000-6d60f4ede681d00297a5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0udi-0900000000-e16ab78095ff95c3688a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-03dl-9800000000-ca8eb82001337c5aa40b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-02t9-9100000000-715895c8385339b110e9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-0900000000-5b91114f7b3c03540b81 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0udi-2900000000-9c18bb28a410b0e40cf2 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0006-9200000000-f7a3946beaf9292bef4d | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0037879 |
|---|
| FooDB ID | FDB017032 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 458437 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 525777 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|