| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 02:24:45 UTC |
|---|
| Update Date | 2016-11-09 01:19:12 UTC |
|---|
| Accession Number | CHEM031128 |
|---|
| Identification |
|---|
| Common Name | Ganolucidic acid E |
|---|
| Class | Small Molecule |
|---|
| Description | Ganolucidic acid E is found in mushrooms. Ganolucidic acid E is a metabolite of Ganoderma lucidum (reishi). |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
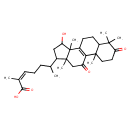
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Ganolucidate e | Generator | | 15alpha-Hydroxy-3,11-dioxo-5alpha-lanosta-8,24E-dien-26-Oic acid | HMDB | | (2Z)-6-{12-hydroxy-2,6,6,11,15-pentamethyl-5,17-dioxotetracyclo[8.7.0.0²,⁷.0¹¹,¹⁵]heptadec-1(10)-en-14-yl}-2-methylhept-2-enoate | Generator |
|
|---|
| Chemical Formula | C30H44O5 |
|---|
| Average Molecular Mass | 484.667 g/mol |
|---|
| Monoisotopic Mass | 484.319 g/mol |
|---|
| CAS Registry Number | 114567-50-9 |
|---|
| IUPAC Name | (2Z)-6-{12-hydroxy-2,6,6,11,15-pentamethyl-5,17-dioxotetracyclo[8.7.0.0²,⁷.0¹¹,¹⁵]heptadec-1(10)-en-14-yl}-2-methylhept-2-enoic acid |
|---|
| Traditional Name | (2Z)-6-{12-hydroxy-2,6,6,11,15-pentamethyl-5,17-dioxotetracyclo[8.7.0.0²,⁷.0¹¹,¹⁵]heptadec-1(10)-en-14-yl}-2-methylhept-2-enoic acid |
|---|
| SMILES | CC(CC\C=C(\C)C(O)=O)C1CC(O)C2(C)C3=C(C(=O)CC12C)C1(C)CCC(=O)C(C)(C)C1CC3 |
|---|
| InChI Identifier | InChI=1S/C30H44O5/c1-17(9-8-10-18(2)26(34)35)20-15-24(33)30(7)19-11-12-22-27(3,4)23(32)13-14-28(22,5)25(19)21(31)16-29(20,30)6/h10,17,20,22,24,33H,8-9,11-16H2,1-7H3,(H,34,35)/b18-10- |
|---|
| InChI Key | XRBLVCACUHPHDE-ZDLGFXPLSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as triterpenoids. These are terpene molecules containing six isoprene units. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Prenol lipids |
|---|
| Sub Class | Triterpenoids |
|---|
| Direct Parent | Triterpenoids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Triterpenoid
- Monohydroxy bile acid, alcohol, or derivatives
- Hydroxy bile acid, alcohol, or derivatives
- Bile acid, alcohol, or derivatives
- Steroid acid
- 3-oxosteroid
- Hydroxysteroid
- 11-oxosteroid
- Oxosteroid
- 15-hydroxysteroid
- Steroid
- Medium-chain fatty acid
- Cyclohexenone
- Hydroxy fatty acid
- Fatty acid
- Fatty acyl
- Unsaturated fatty acid
- Cyclic alcohol
- Ketone
- Secondary alcohol
- Cyclic ketone
- Carboxylic acid
- Carboxylic acid derivative
- Monocarboxylic acid or derivatives
- Organooxygen compound
- Hydrocarbon derivative
- Organic oxygen compound
- Carbonyl group
- Organic oxide
- Alcohol
- Aliphatic homopolycyclic compound
|
|---|
| Molecular Framework | Aliphatic homopolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-014l-0122900000-c00bbbd677c44169ff93 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (2 TMS) - 70eV, Positive | splash10-03di-2121259000-bd45a11dd66ad735ee9b | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014r-0000900000-11f81172f9e30333de4b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-02mv-0103900000-f544812bd14ac56a92d8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-06di-4607900000-10f298ed3057f8e6b8de | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-001i-0000900000-9ded364fed282b24f165 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00sr-0000900000-06f04b00746fbe9b54bf | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0g4l-4020900000-431e5a376ba58c710202 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0002-9202300000-fd30f9fe4fa78b755ed8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0005-9004400000-4b7b7172888b19ec64db | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-014l-9214000000-7bae7931bc1714d2027f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-001i-0000900000-5d9a32665cee1f6c1572 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-01p9-0000900000-e6b10d34a6c949b86420 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-00dr-2006900000-24aec08aa68a1fc06592 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0037607 |
|---|
| FooDB ID | FDB016720 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00023874 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 131752214 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|