| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 02:21:50 UTC |
|---|
| Update Date | 2016-11-09 01:19:11 UTC |
|---|
| Accession Number | CHEM031061 |
|---|
| Identification |
|---|
| Common Name | 6-Hydroxysandoricin |
|---|
| Class | Small Molecule |
|---|
| Description | 6-Hydroxysandoricin is a constituent of Sandoricum koetjape (santol). |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
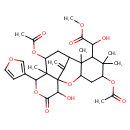
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 2-Amino-3-(4-hydroxyphenyl)propanoic acid | HMDB | | 2-Amino-3-(p-hydroxyphenyl)propionic acid | HMDB | | 3-(p-Hydroxyphenyl)alanine | HMDB | | Tirosina | HMDB | | Tyr | HMDB | | Tyrosin | HMDB | | Y | HMDB | | 2-Amino-3-(4-hydroxyphenyl)propanoate | HMDB | | 2-Amino-3-(p-hydroxyphenyl)propionate | HMDB | | Methyl 2-[5,11-bis(acetyloxy)-13-(furan-3-yl)-16-hydroxy-6,6,8,12-tetramethyl-17-methylidene-15-oxo-2,14-dioxatetracyclo[7.7.1.0¹,¹².0³,⁸]heptadecan-7-yl]-2-hydroxyacetic acid | HMDB | | 6-Hydroxysandoricin | MeSH |
|
|---|
| Chemical Formula | C31H40O12 |
|---|
| Average Molecular Mass | 604.642 g/mol |
|---|
| Monoisotopic Mass | 604.252 g/mol |
|---|
| CAS Registry Number | 133585-56-5 |
|---|
| IUPAC Name | methyl 2-[5,11-bis(acetyloxy)-13-(furan-3-yl)-16-hydroxy-6,6,8,12-tetramethyl-17-methylidene-15-oxo-2,14-dioxatetracyclo[7.7.1.0¹,¹².0³,⁸]heptadecan-7-yl]-2-hydroxyacetate |
|---|
| Traditional Name | methyl 2-[5,11-bis(acetyloxy)-13-(furan-3-yl)-16-hydroxy-6,6,8,12-tetramethyl-17-methylidene-15-oxo-2,14-dioxatetracyclo[7.7.1.0¹,¹².0³,⁸]heptadecan-7-yl]-2-hydroxyacetate |
|---|
| SMILES | COC(=O)C(O)C1C(C)(C)C(CC2OC34C(O)C(=O)OC(C5=COC=C5)C3(C)C(CC(C4=C)C12C)OC(C)=O)OC(C)=O |
|---|
| InChI Identifier | InChI=1S/C31H40O12/c1-14-18-11-21(41-16(3)33)30(7)25(17-9-10-39-13-17)42-27(37)24(35)31(14,30)43-20-12-19(40-15(2)32)28(4,5)23(29(18,20)6)22(34)26(36)38-8/h9-10,13,18-25,34-35H,1,11-12H2,2-8H3 |
|---|
| InChI Key | NKHYHRMLCJNKHW-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as tetracarboxylic acids and derivatives. These are carboxylic acids containing exactly four carboxyl groups. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic acids and derivatives |
|---|
| Class | Carboxylic acids and derivatives |
|---|
| Sub Class | Tetracarboxylic acids and derivatives |
|---|
| Direct Parent | Tetracarboxylic acids and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Tetracarboxylic acid or derivatives
- Delta valerolactone
- Delta_valerolactone
- Oxane
- Furan
- Heteroaromatic compound
- Methyl ester
- Carboxylic acid ester
- Lactone
- Secondary alcohol
- Dialkyl ether
- Ether
- Oxacycle
- Organoheterocyclic compound
- Alcohol
- Organic oxide
- Hydrocarbon derivative
- Carbonyl group
- Organic oxygen compound
- Organooxygen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0005-1000190000-c6508a3f9ac27320a649 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (1 TMS) - 70eV, Positive | splash10-01bc-6001179000-e858323a0b723b25965f | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_2) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_1) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_1_1) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_1_2) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_2_1) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS ("6-Hydroxysandoricin,1TMS,#1" TMS) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0bta-0000291000-87f72b14b31432285cfe | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-000b-0001390000-83be7fbf21a04294b60b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0fdk-3000930000-d405467d2f09b07fb0e5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-11ou-1000092000-7f697eda5cfa153e6c95 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-096u-3000190000-374422525610748d3fca | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0zg0-4000490000-b3836ac2293c69530f69 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0fk9-3000092000-45485af9be02dd0b2027 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4r-6000591000-00cefbd4188c5d414658 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-052b-9000830000-090f666b77f21c34b5f2 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a4i-0000598000-f142dc5fac87d0cea471 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a4r-4001294000-8652e3d66cbaae9b0d6e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0gbc-9000282000-0aacdff2f0d2f8a15395 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0037556 |
|---|
| FooDB ID | FDB016638 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00001397 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Tyrosine |
|---|
| Chemspider ID | 170506 |
|---|
| ChEBI ID | 185400 |
|---|
| PubChem Compound ID | 196858 |
|---|
| Kegg Compound ID | C01536 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|