| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 02:20:28 UTC |
|---|
| Update Date | 2016-11-09 01:19:11 UTC |
|---|
| Accession Number | CHEM031032 |
|---|
| Identification |
|---|
| Common Name | Galanolactone |
|---|
| Class | Small Molecule |
|---|
| Description | Galanolactone is found in ginger. Galanolactone is a constituent of Alpinia galanga (greater galangal) Galanolactone is a diterpenoid lactone first isolated from ginger. It is known to be present in acetone extracts of ginger, and appears to be an antagonist at 5-HT3 receptors. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
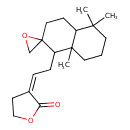
| Chemical Structure | |
|---|
| Synonyms | Not Available |
|---|
| Chemical Formula | C20H30O3 |
|---|
| Average Molecular Mass | 318.450 g/mol |
|---|
| Monoisotopic Mass | 318.219 g/mol |
|---|
| CAS Registry Number | 115753-79-2 |
|---|
| IUPAC Name | (3Z)-3-(2-{5,5,8a-trimethyl-octahydro-1H-spiro[naphthalene-2,2'-oxirane]-1-yl}ethylidene)oxolan-2-one |
|---|
| Traditional Name | (3Z)-3-(2-{5,5,8a-trimethyl-hexahydro-1H-spiro[naphthalene-2,2'-oxirane]-1-yl}ethylidene)oxolan-2-one |
|---|
| SMILES | CC1(C)CCCC2(C)C(C\C=C3\CCOC3=O)C3(CO3)CCC12 |
|---|
| InChI Identifier | InChI=1S/C20H30O3/c1-18(2)9-4-10-19(3)15(18)7-11-20(13-23-20)16(19)6-5-14-8-12-22-17(14)21/h5,15-16H,4,6-13H2,1-3H3/b14-5- |
|---|
| InChI Key | MBPTXJNHCBXMBP-RZNTYIFUSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as gamma butyrolactones. Gamma butyrolactones are compounds containing a gamma butyrolactone moiety, which consists of an aliphatic five-member ring with four carbon atoms, one oxygen atom, and bears a ketone group on the carbon adjacent to the oxygen atom. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Lactones |
|---|
| Sub Class | Gamma butyrolactones |
|---|
| Direct Parent | Gamma butyrolactones |
|---|
| Alternative Parents | |
|---|
| Substituents | - Gamma butyrolactone
- Tetrahydrofuran
- Enoate ester
- Alpha,beta-unsaturated carboxylic ester
- Carboxylic acid ester
- Carboxylic acid derivative
- Dialkyl ether
- Oxirane
- Ether
- Monocarboxylic acid or derivatives
- Oxacycle
- Organic oxygen compound
- Organic oxide
- Carbonyl group
- Organooxygen compound
- Hydrocarbon derivative
- Aliphatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aliphatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0f79-0691000000-76ff2be2a471bb872fd2 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014i-1079000000-1752926143446e156771 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0fsj-2192000000-34665ab35024bcaec03b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0udi-8920000000-9070c224eeba82a073c6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-014i-1019000000-c8e6f08fa9a4c772c41e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-01b9-3095000000-9f4bfe9957ad918fccc7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0pb9-4090000000-bec7fcb4c3d0c3bfeb83 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014i-0009000000-f44b8f78f028f8f8f3b6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0g29-0913000000-29e7ab868d79d3e36a1a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0fi0-6923000000-8d5099da71cc9b483052 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-014i-0009000000-062ad2a35e11d303cf23 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-014i-0029000000-eefd64fbf57a97fa197a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-2091000000-300e0967ce3af0cdcdf5 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0037523 |
|---|
| FooDB ID | FDB016604 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00034848 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Galanolactone |
|---|
| Chemspider ID | 35014427 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 57457805 |
|---|
| Kegg Compound ID | C17498 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|