| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 02:19:46 UTC |
|---|
| Update Date | 2016-11-09 01:19:11 UTC |
|---|
| Accession Number | CHEM031017 |
|---|
| Identification |
|---|
| Common Name | 3,3',5,7-Tetrahydroxy-4'-methoxyflavanone |
|---|
| Class | Small Molecule |
|---|
| Description | Isolated from Blumea balsamifera (sambong). 3,3',5,7-Tetrahydroxy-4'-methoxyflavanone is found in tea. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
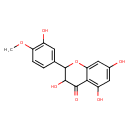
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Dihydrotamarixetin | HMDB | | 3,3',5,7-Tetrahydroxy-4'-methoxyflavone | HMDB |
|
|---|
| Chemical Formula | C16H14O7 |
|---|
| Average Molecular Mass | 318.278 g/mol |
|---|
| Monoisotopic Mass | 318.074 g/mol |
|---|
| CAS Registry Number | 70411-27-7 |
|---|
| IUPAC Name | 3,5,7-trihydroxy-2-(3-hydroxy-4-methoxyphenyl)-3,4-dihydro-2H-1-benzopyran-4-one |
|---|
| Traditional Name | quercetin 4'-methyl ether |
|---|
| SMILES | COC1=C(O)C=C(C=C1)C1OC2=CC(O)=CC(O)=C2C(=O)C1O |
|---|
| InChI Identifier | InChI=1S/C16H14O7/c1-22-11-3-2-7(4-9(11)18)16-15(21)14(20)13-10(19)5-8(17)6-12(13)23-16/h2-6,15-19,21H,1H3 |
|---|
| InChI Key | KQNGHARGJDXHKF-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as 4'-o-methylated flavonoids. These are flavonoids with methoxy groups attached to the C4' atom of the flavonoid backbone. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Phenylpropanoids and polyketides |
|---|
| Class | Flavonoids |
|---|
| Sub Class | O-methylated flavonoids |
|---|
| Direct Parent | 4'-O-methylated flavonoids |
|---|
| Alternative Parents | |
|---|
| Substituents | - 4p-methoxyflavonoid-skeleton
- 3'-hydroxyflavonoid
- 3-hydroxyflavonoid
- 5-hydroxyflavonoid
- 7-hydroxyflavonoid
- Flavanone
- Flavanonol
- Hydroxyflavonoid
- Flavan
- Chromone
- Chromane
- Benzopyran
- Methoxyphenol
- 1-benzopyran
- Phenoxy compound
- Anisole
- Methoxybenzene
- Aryl alkyl ketone
- Aryl ketone
- Phenol ether
- Phenol
- Alkyl aryl ether
- 1-hydroxy-2-unsubstituted benzenoid
- 1-hydroxy-4-unsubstituted benzenoid
- Benzenoid
- Monocyclic benzene moiety
- Vinylogous acid
- Ketone
- Secondary alcohol
- Polyol
- Organoheterocyclic compound
- Ether
- Oxacycle
- Organic oxide
- Organooxygen compound
- Organic oxygen compound
- Alcohol
- Hydrocarbon derivative
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-000i-0942000000-243d67f1bef64e8ec397 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (4 TMS) - 70eV, Positive | splash10-002f-2430090000-e78153eb3884bd8edb1a | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014i-0109000000-214294e6786df702f543 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0uxr-0925000000-96df061b2c051eba81fb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0uki-3900000000-986176e74a8e3aed42ec | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-014i-0309000000-2c0940e32c2ac06d9454 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0gb9-0925000000-80ccd9f800335c4efcf3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0pbl-3910000000-95fddf0306ab882ab5a7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-014i-0009000000-bf47ad651d2af1b09144 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-014i-0309000000-4f56e4ee0972dae05af2 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a59-2900000000-262acfca5032dda41efe | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014i-0509000000-07dd9e06c9a656c5cb4b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0gdi-0906000000-1601e6e6e99b2c1666eb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0udi-0900000000-97b177f72d54ce0fe519 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0002937 |
|---|
| FooDB ID | FDB016577 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00004636 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | 3446 |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Tamarixetin |
|---|
| Chemspider ID | 423348 |
|---|
| ChEBI ID | 542812 |
|---|
| PubChem Compound ID | 482576 |
|---|
| Kegg Compound ID | C10188 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | | 1. Rao, Koppaka V.; Owoyale, Jacob A. Partial methylation of quercetin: direct synthesis of tamarixetin, ombuin and ayanin. Journal of Heterocyclic Chemistry (1976), 13(6), 1293-5. | | 2. Nielsen SE, Freese R, Kleemola P, Mutanen M: Flavonoids in human urine as biomarkers for intake of fruits and vegetables. Cancer Epidemiol Biomarkers Prev. 2002 May;11(5):459-66. | | 3. Brevik A, Rasmussen SE, Drevon CA, Andersen LF: Urinary excretion of flavonoids reflects even small changes in the dietary intake of fruits and vegetables. Cancer Epidemiol Biomarkers Prev. 2004 May;13(5):843-9. | | 4. Wu, Z., Smith, J. V., Paramasivam, V., Butko, P., Khan, I., Cypser, J. R., and Luo, Y. (2002). Ginkgo biloba extract EGb 761 increases stress resistance and extends life span of caenoraibditis elegans. Cell Mol Biol (Noisy-le-grand) 48, 725-31. |
|
|---|