| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 02:04:45 UTC |
|---|
| Update Date | 2016-11-09 01:19:07 UTC |
|---|
| Accession Number | CHEM030679 |
|---|
| Identification |
|---|
| Common Name | Istanbulin A |
|---|
| Class | Small Molecule |
|---|
| Description | Istanbulin A is found in green vegetables. Istanbulin A is isolated from Smyrnium olusatrum (alexanders). |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
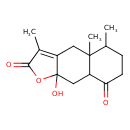
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| ent-8a-Hydroxy-1-oxo-7(11)-eremophilen-12,8-olide | HMDB | | Istanbulin-a | HMDB, MeSH | | Istanbulin a | MeSH |
|
|---|
| Chemical Formula | C15H20O4 |
|---|
| Average Molecular Mass | 264.317 g/mol |
|---|
| Monoisotopic Mass | 264.136 g/mol |
|---|
| CAS Registry Number | 35481-83-5 |
|---|
| IUPAC Name | 9a-hydroxy-3,4a,5-trimethyl-2H,4H,4aH,5H,6H,7H,8H,8aH,9H,9aH-naphtho[2,3-b]furan-2,8-dione |
|---|
| Traditional Name | 9a-hydroxy-3,4a,5-trimethyl-4H,5H,6H,7H,8aH,9H-naphtho[2,3-b]furan-2,8-dione |
|---|
| SMILES | CC1CCC(=O)C2CC3(O)OC(=O)C(C)=C3CC12C |
|---|
| InChI Identifier | InChI=1S/C15H20O4/c1-8-4-5-12(16)11-7-15(18)10(6-14(8,11)3)9(2)13(17)19-15/h8,11,18H,4-7H2,1-3H3 |
|---|
| InChI Key | HYBIDSYXKTYHCM-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as terpene lactones. These are prenol lipids containing a lactone ring. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Prenol lipids |
|---|
| Sub Class | Terpene lactones |
|---|
| Direct Parent | Terpene lactones |
|---|
| Alternative Parents | |
|---|
| Substituents | - Terpene lactone
- Eremophilanolide or secoeremophilanolide
- Sesquiterpenoid
- Naphthofuran
- 2-furanone
- Cyclic alcohol
- Dihydrofuran
- Enoate ester
- Alpha,beta-unsaturated carboxylic ester
- Lactone
- Ketone
- Hemiacetal
- Carboxylic acid ester
- Monocarboxylic acid or derivatives
- Oxacycle
- Organoheterocyclic compound
- Carboxylic acid derivative
- Organooxygen compound
- Hydrocarbon derivative
- Organic oxide
- Organic oxygen compound
- Carbonyl group
- Aliphatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aliphatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-000i-1960000000-61907d8d15b1cbc582d5 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (1 TMS) - 70eV, Positive | splash10-009i-8594000000-e3d285e622927e744842 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014i-0390000000-4bbdc72d7be928afeddd | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00ke-2960000000-2441ad518e158dd2ba9f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0k97-9300000000-ea930d4bee3a28712293 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03di-0090000000-ca8ed823a3d7b408e00c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0900-0090000000-2beb80d806f8e4bfb86b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-00l6-5690000000-c385f2ffc88d58522478 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03di-0090000000-16d43704d08f3756e03a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-03di-0090000000-cf4dd69ba3cf077e83ed | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-03ki-2890000000-0da57c6995395368a5c2 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014i-0090000000-8d9e65027e815d92f341 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-014i-0290000000-dc1ee00146a9b0d0427c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-00si-3920000000-48bfbf4c83ccd0e454ab | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0037055 |
|---|
| FooDB ID | FDB016038 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00017304 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 35014361 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 12304519 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|