| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 01:59:11 UTC |
|---|
| Update Date | 2016-11-09 01:19:06 UTC |
|---|
| Accession Number | CHEM030559 |
|---|
| Identification |
|---|
| Common Name | Gibberellin A38 glucosyl ester |
|---|
| Class | Small Molecule |
|---|
| Description | Not Available |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
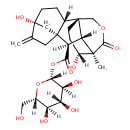
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (2R,3S,4R,5R,6S)-3,4,5-Trihydroxy-6-(hydroxymethyl)oxan-2-yl (1S,2S,5R,8R,9R,10R,11R,17R)-5,17-dihydroxy-11-methyl-6-methylidene-12-oxo-13-oxapentacyclo[9.3.3.1,.0,.0,]octadecane-9-carboxylic acid | Generator | | (2R,3S,4R,5R,6S)-3,4,5-Trihydroxy-6-(hydroxymethyl)oxan-2-yl (1S,2S,5R,8R,9R,10R,11R,17R)-5,17-dihydroxy-11-methyl-6-methylidene-12-oxo-13-oxapentacyclo[9.3.3.1⁵,⁸.0¹,¹⁰.0²,⁸]octadecane-9-carboxylic acid | Generator |
|
|---|
| Chemical Formula | C26H36O11 |
|---|
| Average Molecular Mass | 524.563 g/mol |
|---|
| Monoisotopic Mass | 524.226 g/mol |
|---|
| CAS Registry Number | 36702-73-5 |
|---|
| IUPAC Name | (2R,3S,4R,5R,6S)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl (1S,2S,5R,8R,9R,10R,11R,17R)-5,17-dihydroxy-11-methyl-6-methylidene-12-oxo-13-oxapentacyclo[9.3.3.1⁵,⁸.0¹,¹⁰.0²,⁸]octadecane-9-carboxylate |
|---|
| Traditional Name | (2R,3S,4R,5R,6S)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl (1S,2S,5R,8R,9R,10R,11R,17R)-5,17-dihydroxy-11-methyl-6-methylidene-12-oxo-13-oxapentacyclo[9.3.3.1⁵,⁸.0¹,¹⁰.0²,⁸]octadecane-9-carboxylate |
|---|
| SMILES | [H][C@]12CC[C@@]3(O)C[C@@]1(CC3=C)[C@]([H])(C(=O)O[C@@]1([H])O[C@@]([H])(CO)[C@]([H])(O)[C@@]([H])(O)[C@]1([H])O)[C@]1([H])[C@]22CC[C@@]([H])(O)[C@]1(C)C(=O)OC2 |
|---|
| InChI Identifier | InChI=1S/C26H36O11/c1-11-7-25-9-26(11,34)6-3-13(25)24-5-4-14(28)23(2,22(33)35-10-24)19(24)15(25)20(32)37-21-18(31)17(30)16(29)12(8-27)36-21/h12-19,21,27-31,34H,1,3-10H2,2H3/t12-,13+,14+,15-,16-,17+,18-,19-,21+,23-,24-,25+,26+/m0/s1 |
|---|
| InChI Key | DDEBJBVQDFATPL-SAYSAXKISA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as diterpene glycosides. These are diterpenoids in which an isoprene unit is glycosylated. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Prenol lipids |
|---|
| Sub Class | Terpene glycosides |
|---|
| Direct Parent | Diterpene glycosides |
|---|
| Alternative Parents | |
|---|
| Substituents | - Diterpene glycoside
- Diterpenoid
- Diterpene lactone
- 20-norgibberellane diterpenoid
- Gibberellin
- Fatty acyl glycoside of mono- or disaccharide
- Fatty acyl glycoside
- Hexose monosaccharide
- Delta_valerolactone
- Delta valerolactone
- Fatty acyl
- Oxane
- Monosaccharide
- Dicarboxylic acid or derivatives
- Tertiary alcohol
- Cyclic alcohol
- Secondary alcohol
- Lactone
- Carboxylic acid ester
- Oxacycle
- Organoheterocyclic compound
- Polyol
- Carboxylic acid derivative
- Acetal
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Primary alcohol
- Organooxygen compound
- Carbonyl group
- Alcohol
- Aliphatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aliphatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-06tb-0209140000-5ccae0da672abef1f873 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0002-0309110000-29e3b98efa63175c86c8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0002-2429000000-4e68ed9a80881e42b0e4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-01vo-0109250000-7cdd556cee7deb4d4a02 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-03dl-2309110000-b81d07baff54352bbfc2 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-01ox-7209000000-1d1112a28c286c0155c1 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | Not Available |
|---|
| FooDB ID | Not Available |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | Not Available |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|