| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 01:58:40 UTC |
|---|
| Update Date | 2016-11-09 01:19:05 UTC |
|---|
| Accession Number | CHEM030550 |
|---|
| Identification |
|---|
| Common Name | Gibberellin A19 |
|---|
| Class | Small Molecule |
|---|
| Description | Constituent of moso bamboo shoots (Phyllostachys edulis). Gibberellin A19 is found in many foods, some of which are swede, devilfish, vanilla, and canola. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
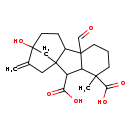
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Bamboo gibberellin | HMDB | | GA19 | HMDB | | GA19 CPD | HMDB | | Gibberellin 19 | HMDB | | Gibberellin a(19) | HMDB | | Gibberellin ga19 | HMDB |
|
|---|
| Chemical Formula | C20H26O6 |
|---|
| Average Molecular Mass | 362.417 g/mol |
|---|
| Monoisotopic Mass | 362.173 g/mol |
|---|
| CAS Registry Number | 6980-44-5 |
|---|
| IUPAC Name | 8-formyl-12-hydroxy-4-methyl-13-methylidenetetracyclo[10.2.1.0^{1,9}.0^{3,8}]pentadecane-2,4-dicarboxylic acid |
|---|
| Traditional Name | 8-formyl-12-hydroxy-4-methyl-13-methylidenetetracyclo[10.2.1.0^{1,9}.0^{3,8}]pentadecane-2,4-dicarboxylic acid |
|---|
| SMILES | [H][C@@]12CC[C@]3(O)C[C@]1(CC3=C)[C@@H](C(O)=O)[C@@]1([H])[C@]2(CCC[C@@]1(C)C(O)=O)C=O |
|---|
| InChI Identifier | InChI=1S/C20H26O6/c1-11-8-19-9-20(11,26)7-4-12(19)18(10-21)6-3-5-17(2,16(24)25)14(18)13(19)15(22)23/h10,12-14,26H,1,3-9H2,2H3,(H,22,23)(H,24,25)/t12-,13+,14+,17+,18+,19-,20-/m0/s1 |
|---|
| InChI Key | VNCQCPQAMDQEBY-YTJHIPEWSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as c20-gibberellin 6-carboxylic acids. These are c20-gibberellins with a carboxyl group at the 6-position. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Prenol lipids |
|---|
| Sub Class | Diterpenoids |
|---|
| Direct Parent | C20-gibberellin 6-carboxylic acids |
|---|
| Alternative Parents | Not Available |
|---|
| Substituents | Not Available |
|---|
| Molecular Framework | Aliphatic homopolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-00ls-1209000000-4c2a3285093a0c4c8473 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (3 TMS) - 70eV, Positive | splash10-03di-4010590000-795da2ada8bd05fb94eb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0002-0019000000-d04f66a3669562a1a3ca | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0002-0359000000-a5f21b0d106847946431 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-006t-1394000000-723214d3e0597593b21f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03xr-0019000000-68cd0d21d27d35c1d7f9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-01ba-0069000000-4d84555a53b776e6b21e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-00y1-0194000000-f0d1b338bfd488d55664 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0036896 |
|---|
| FooDB ID | FDB015856 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00000019 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 3822439 |
|---|
| ChEBI ID | 28731 |
|---|
| PubChem Compound ID | 4632015 |
|---|
| Kegg Compound ID | C02034 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|