| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 01:57:55 UTC |
|---|
| Update Date | 2016-11-09 01:19:05 UTC |
|---|
| Accession Number | CHEM030536 |
|---|
| Identification |
|---|
| Common Name | Reticulataxanthin |
|---|
| Class | Small Molecule |
|---|
| Description | Reticulataxanthin is found in citrus. Peel constituent of tangerine (Citrus reticulata) and the tangerine hybrid (Minneola tangor). |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
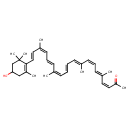
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 3-Hydroxy-6'-methyl-6'-apo-b-caroten-6'-one | HMDB | | 3-Hydroxycitraniaxanthin | HMDB | | Reticulaxanthin | HMDB |
|
|---|
| Chemical Formula | C33H44O2 |
|---|
| Average Molecular Mass | 472.701 g/mol |
|---|
| Monoisotopic Mass | 472.334 g/mol |
|---|
| CAS Registry Number | 28368-09-4 |
|---|
| IUPAC Name | (3Z,5E,7Z,9E,11E,13Z,15E,17Z,19E)-20-(4-hydroxy-2,6,6-trimethylcyclohex-1-en-1-yl)-5,9,14,18-tetramethylicosa-3,5,7,9,11,13,15,17,19-nonaen-2-one |
|---|
| Traditional Name | (3Z,5E,7Z,9E,11E,13Z,15E,17Z,19E)-20-(4-hydroxy-2,6,6-trimethylcyclohex-1-en-1-yl)-5,9,14,18-tetramethylicosa-3,5,7,9,11,13,15,17,19-nonaen-2-one |
|---|
| SMILES | CC(=O)\C=C/C(/C)=C/C=C\C(\C)=C\C=C\C=C(\C)/C=C/C=C(/C)\C=C\C1=C(C)CC(O)CC1(C)C |
|---|
| InChI Identifier | InChI=1S/C33H44O2/c1-25(15-11-17-27(3)19-21-30(6)34)13-9-10-14-26(2)16-12-18-28(4)20-22-32-29(5)23-31(35)24-33(32,7)8/h9-22,31,35H,23-24H2,1-8H3/b10-9+,15-11-,16-12+,21-19-,22-20+,25-13+,26-14-,27-17+,28-18- |
|---|
| InChI Key | JNRFHJQRIUJTNO-DBTGPYIJSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as triterpenoids. These are terpene molecules containing six isoprene units. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Prenol lipids |
|---|
| Sub Class | Triterpenoids |
|---|
| Direct Parent | Triterpenoids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Triterpenoid
- Alpha,beta-unsaturated ketone
- Enone
- Acryloyl-group
- Secondary alcohol
- Ketone
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Carbonyl group
- Alcohol
- Aliphatic homomonocyclic compound
|
|---|
| Molecular Framework | Aliphatic homomonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0a4i-1000900000-029525b772db4f6f1f85 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (1 TMS) - 70eV, Positive | splash10-004i-5110290000-bfced86cd4f4f0243363 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0ab9-0111900000-b3177155263c6706e52a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0uy1-0559500000-eb1d498ba0a322a56436 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0f6t-2569100000-ae8e8ffd7a8ca2338ac4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-00di-0000900000-e6d03a734023c3234c60 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0fk9-0000900000-550f1db68b08dd11cfcc | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0pbi-1221900000-1185a69e36e646740726 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-00fr-0119700000-78040f9b610b4bb07df4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-05fu-8708900000-377bc86a5f4bd99cf238 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-000j-4419300000-51e10039c77f7d8043ad | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0gi0-0133900000-cb99794c8bc1096ac0a5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0f92-1309500000-0379dd51e0714fc62fc0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-01s9-2965200000-2dcaa72b7399add3ada9 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0036882 |
|---|
| FooDB ID | FDB015840 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00057325 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 35014294 |
|---|
| ChEBI ID | 176158 |
|---|
| PubChem Compound ID | 131752077 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|