| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 01:57:21 UTC |
|---|
| Update Date | 2016-11-09 01:19:05 UTC |
|---|
| Accession Number | CHEM030522 |
|---|
| Identification |
|---|
| Common Name | Cryptoflavin |
|---|
| Class | Small Molecule |
|---|
| Description | Cryptoflavin is found in citrus. Cryptoflavin is a constituent of persimmon (Diospyros kaki), star fruit (Averrhoa carambola), orange (Citrus sinensis) and alfalfa (Medicago) species Poss. isolated from peach (Prunus persica). |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
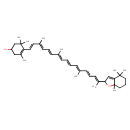
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 5,8-Epoxy-5,8-dihydro-b,b-caroten-3-ol | HMDB | | Cryptoxanthin 5,8-epoxide | HMDB |
|
|---|
| Chemical Formula | C40H56O2 |
|---|
| Average Molecular Mass | 568.871 g/mol |
|---|
| Monoisotopic Mass | 568.428 g/mol |
|---|
| CAS Registry Number | 30311-63-8 |
|---|
| IUPAC Name | 4-[(1E,3E,5E,7E,9E,11E,13E,15E)-16-(4,4,7a-trimethyl-2,4,5,6,7,7a-hexahydro-1-benzofuran-2-yl)-3,7,12-trimethylheptadeca-1,3,5,7,9,11,13,15-octaen-1-yl]-3,5,5-trimethylcyclohex-3-en-1-ol |
|---|
| Traditional Name | 4-[(1E,3E,5E,7E,9E,11E,13E,15E)-16-(4,4,7a-trimethyl-2,5,6,7-tetrahydro-1-benzofuran-2-yl)-3,7,12-trimethylheptadeca-1,3,5,7,9,11,13,15-octaen-1-yl]-3,5,5-trimethylcyclohex-3-en-1-ol |
|---|
| SMILES | C\C(\C=C\C=C(/C)\C=C\C1=C(C)CC(O)CC1(C)C)=C/C=C/C=C(\C)/C=C/C=C(\C)C1OC2(C)CCCC(C)(C)C2=C1 |
|---|
| InChI Identifier | InChI=1S/C40H56O2/c1-29(18-13-19-31(3)22-23-35-33(5)26-34(41)28-39(35,8)9)16-11-12-17-30(2)20-14-21-32(4)36-27-37-38(6,7)24-15-25-40(37,10)42-36/h11-14,16-23,27,34,36,41H,15,24-26,28H2,1-10H3/b12-11+,18-13+,20-14+,23-22+,29-16+,30-17+,31-19+,32-21+ |
|---|
| InChI Key | WEJIOGMJJWSQFC-NNAJIMERSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as xanthophylls. These are carotenoids containing an oxygenated carotene backbone. Carotenes are characterized by the presence of two end-groups (mostly cyclohexene rings, but also cyclopentene rings or acyclic groups) linked by a long branched alkyl chain. Carotenes belonging form a subgroup of the carotenoids family. Xanthophylls arise by oxygenation of the carotene backbone. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Prenol lipids |
|---|
| Sub Class | Tetraterpenoids |
|---|
| Direct Parent | Xanthophylls |
|---|
| Alternative Parents | |
|---|
| Substituents | - Xanthophyll
- Benzofuran
- Dihydrofuran
- Secondary alcohol
- Oxacycle
- Organoheterocyclic compound
- Ether
- Dialkyl ether
- Organic oxygen compound
- Hydrocarbon derivative
- Organooxygen compound
- Alcohol
- Aliphatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aliphatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0udi-1400190000-1eeb767d7c5d027d14cf | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (1 TMS) - 70eV, Positive | splash10-004i-2500019000-078544e499aca653934a | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS ("Cryptoflavin,1TMS,#1" TMS) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_1_1) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0gb9-0442390000-ac9d4c98eec7bb83d3b2 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-014i-0898710000-20869f6e205b7aba60f7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0670-5978720000-640a9e931e4989356ea9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-014i-0000090000-99fe9ab8c38116dbbf99 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-014i-0300090000-45e895abd2dc31c12205 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0udi-0923560000-f08a58a94ebc282aea2e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-014i-0000090000-7d3a81b5730e4ceb4ecd | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-014i-0124190000-930c8bb34f502dc35257 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-005j-0169210000-f412665c7736f1994da1 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014i-0174590000-c55da7adae2523e26519 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-001a-0279350000-a0f38b9bd8e83d0aa771 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0002-0559000000-bef445a32fed6aa655f1 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0036870 |
|---|
| FooDB ID | FDB015825 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00054617 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 4525635 |
|---|
| ChEBI ID | 176090 |
|---|
| PubChem Compound ID | 5376350 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|