| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 01:54:12 UTC |
|---|
| Update Date | 2016-11-09 01:19:04 UTC |
|---|
| Accession Number | CHEM030453 |
|---|
| Identification |
|---|
| Common Name | Azorhodine 2G |
|---|
| Class | Small Molecule |
|---|
| Description | Colourant additive for food. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
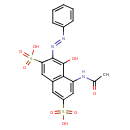
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 5-(acetylamino)-4-Hydroxy-3-(phenylazo)-2,7-naphthalenedisulfonic acid, 9ci | HMDB | | Brilliant acid red g | HMDB | | e128 | HMDB | | Fast crimson GR | HMDB | | Naphthazine rose 2g | HMDB | | Red 2g | HMDB | | Solar fast red 3g | HMDB | | Vopsider red astr g | HMDB | | N-{8-hydroxy-7-[(e)-2-phenyldiazen-1-yl]-3,6-disulfonaphthalen-1-yl}ethanimidate | Generator | | N-{8-hydroxy-7-[(e)-2-phenyldiazen-1-yl]-3,6-disulphonaphthalen-1-yl}ethanimidate | Generator | | N-{8-hydroxy-7-[(e)-2-phenyldiazen-1-yl]-3,6-disulphonaphthalen-1-yl}ethanimidic acid | Generator |
|
|---|
| Chemical Formula | C18H15N3O8S2 |
|---|
| Average Molecular Mass | 465.457 g/mol |
|---|
| Monoisotopic Mass | 465.030 g/mol |
|---|
| CAS Registry Number | Not Available |
|---|
| IUPAC Name | 5-acetamido-4-hydroxy-3-[(E)-2-phenyldiazen-1-yl]naphthalene-2,7-disulfonic acid |
|---|
| Traditional Name | 5-acetamido-4-hydroxy-3-[(E)-2-phenyldiazen-1-yl]naphthalene-2,7-disulfonic acid |
|---|
| SMILES | CC(=O)NC1=CC(=CC2=C1C(O)=C(\N=N\C1=CC=CC=C1)C(=C2)S(O)(=O)=O)S(O)(=O)=O |
|---|
| InChI Identifier | InChI=1S/C18H15N3O8S2/c1-10(22)19-14-9-13(30(24,25)26)7-11-8-15(31(27,28)29)17(18(23)16(11)14)21-20-12-5-3-2-4-6-12/h2-9,23H,1H3,(H,19,22)(H,24,25,26)(H,27,28,29)/b21-20+ |
|---|
| InChI Key | RSNSKUBBVCGSND-QZQOTICOSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as 2-naphthalene sulfonates. These are organic aromatic compounds that contain a naphthalene moiety that carries a sulfonic acid group at the 2-position. Naphthalene is a bicyclic compound that is made up of two fused benzene ring. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Benzenoids |
|---|
| Class | Naphthalenes |
|---|
| Sub Class | Naphthalene sulfonic acids and derivatives |
|---|
| Direct Parent | 2-naphthalene sulfonates |
|---|
| Alternative Parents | |
|---|
| Substituents | - 2-naphthalene sulfonic acid or derivatives
- 2-naphthalene sulfonate
- 1-naphthol
- Arylsulfonic acid or derivatives
- N-acetylarylamine
- 1-sulfo,2-unsubstituted aromatic compound
- N-arylamide
- 1-hydroxy-4-unsubstituted benzenoid
- Monocyclic benzene moiety
- Organic sulfonic acid or derivatives
- Organosulfonic acid or derivatives
- Organosulfonic acid
- Sulfonyl
- Acetamide
- Azo compound
- Secondary carboxylic acid amide
- Carboxamide group
- Carboxylic acid derivative
- Organic 1,3-dipolar compound
- Propargyl-type 1,3-dipolar organic compound
- Organic oxygen compound
- Organic nitrogen compound
- Organosulfur compound
- Organooxygen compound
- Organonitrogen compound
- Organopnictogen compound
- Organic oxide
- Hydrocarbon derivative
- Carbonyl group
- Aromatic homopolycyclic compound
|
|---|
| Molecular Framework | Aromatic homopolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0006-3009300000-0ed85995936be97ba87c | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (1 TMS) - 70eV, Positive | splash10-06xx-3000910000-564e0842a9b0cc2525ba | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-01ba-1002900000-abb975963a264fb7d003 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00ec-1009700000-f3b4fa22c7aacce24b6d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0f6x-4029000000-8ee7a32f640121b452d9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03di-3004900000-81f4c089e66452600f9c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-007o-6019600000-8ee970efdc6b584da8d1 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-000x-9012000000-acd6d77f15fe01d396f4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014i-0000900000-380cd3ebb3857b9aad89 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-014i-0001900000-4b2a75d352382fe87c7e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-014l-7739100000-4e5c4801f62424b529f3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03di-0000900000-77570c9e9bdcff21ef1c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-03di-1001900000-ebfe8b02e3814ee037a2 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0006-9002000000-dd4a344d91f1da38bbda | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0036794 |
|---|
| FooDB ID | FDB015740 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 14731274 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | Not Available |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|