| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 01:50:13 UTC |
|---|
| Update Date | 2016-11-09 01:19:03 UTC |
|---|
| Accession Number | CHEM030348 |
|---|
| Identification |
|---|
| Common Name | Millefin |
|---|
| Class | Small Molecule |
|---|
| Description | Millefin is found in herbs and spices. Millefin is a constituent of Achillea millefolium (yarrow). |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
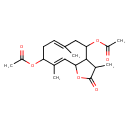
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 9-(Acetyloxy)-3,6,10-trimethyl-2-oxo-2H,3H,3ah,4H,5H,8H,9H,11ah-cyclodeca[b]furan-4-yl acetic acid | HMDB |
|
|---|
| Chemical Formula | C19H26O6 |
|---|
| Average Molecular Mass | 350.406 g/mol |
|---|
| Monoisotopic Mass | 350.173 g/mol |
|---|
| CAS Registry Number | 39262-27-6 |
|---|
| IUPAC Name | 4-(acetyloxy)-3,6,10-trimethyl-2-oxo-2H,3H,3aH,4H,5H,8H,9H,11aH-cyclodeca[b]furan-9-yl acetate |
|---|
| Traditional Name | 4-(acetyloxy)-3,6,10-trimethyl-2-oxo-3H,3aH,4H,5H,8H,9H,11aH-cyclodeca[b]furan-9-yl acetate |
|---|
| SMILES | CC1C2C(OC1=O)\C=C(C)\C(C\C=C(C)\CC2OC(C)=O)OC(C)=O |
|---|
| InChI Identifier | InChI=1S/C19H26O6/c1-10-6-7-15(23-13(4)20)11(2)9-17-18(12(3)19(22)25-17)16(8-10)24-14(5)21/h6,9,12,15-18H,7-8H2,1-5H3/b10-6+,11-9+ |
|---|
| InChI Key | OHMAVTDVTQMMMR-BBYAVRKXSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as germacranolides and derivatives. These are sesquiterpene lactones with a structure based on the germacranolide skeleton, characterized by a gamma lactone fused to a 1,7-dimethylcyclodec-1-ene moiety. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Prenol lipids |
|---|
| Sub Class | Terpene lactones |
|---|
| Direct Parent | Germacranolides and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Germacranolide
- Germacrane sesquiterpenoid
- Sesquiterpenoid
- Tricarboxylic acid or derivatives
- Gamma butyrolactone
- Tetrahydrofuran
- Carboxylic acid ester
- Lactone
- Carboxylic acid derivative
- Oxacycle
- Organoheterocyclic compound
- Hydrocarbon derivative
- Organic oxide
- Organic oxygen compound
- Carbonyl group
- Organooxygen compound
- Aliphatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aliphatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0007-9071000000-2d81073fd33e45c69675 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0zfu-0059000000-684eb1cd405f0501e6be | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-05no-2092000000-49f1f30107d2a5ba10f3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0gb9-9460000000-04e5d862d86a411b5b91 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4j-0029000000-2499d7c09ef73fa8e6c5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4i-3079000000-cabc338d263d13f77f31 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0k9l-9281000000-365b36d23147562fc721 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-000x-0090000000-b5c1a9e6e991867cc16e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-003s-0090000000-8160e6e7436496f70a92 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-004i-1090000000-0fbd709be7add85fcaa1 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0002-0096000000-5c79319a6e925ab232ce | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4l-9031000000-630d1c8ba3621eff899b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4l-9000000000-560c36bc1aba47465cbb | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0036689 |
|---|
| FooDB ID | FDB015619 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00011756 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 4478045 |
|---|
| ChEBI ID | 175476 |
|---|
| PubChem Compound ID | 5319830 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|