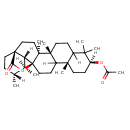| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 01:49:42 UTC |
|---|
| Update Date | 2016-11-09 01:19:03 UTC |
|---|
| Accession Number | CHEM030334 |
|---|
| Identification |
|---|
| Common Name | Ursololactone |
|---|
| Class | Small Molecule |
|---|
| Description | Ursololactone is found in herbs and spices. Ursololactone is isolated from Helichrysum italicum (curry plant). |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 3b-Acetoxy-28,13-ursanolide | HMDB | | (1S,4S,5R,10S,13R,17S,18R,19S,20R)-4,5,9,9,13,19,20-Heptamethyl-23-oxo-24-oxahexacyclo[15.5.2.0¹,¹⁸.0⁴,¹⁷.0⁵,¹⁴.0⁸,¹³]tetracosan-10-yl acetic acid | Generator |
|
|---|
| Chemical Formula | C32H50O4 |
|---|
| Average Molecular Mass | 498.737 g/mol |
|---|
| Monoisotopic Mass | 498.371 g/mol |
|---|
| CAS Registry Number | 28290-51-9 |
|---|
| IUPAC Name | (1S,4S,5R,10S,13R,17S,18R,19S,20R)-4,5,9,9,13,19,20-heptamethyl-23-oxo-24-oxahexacyclo[15.5.2.0¹,¹⁸.0⁴,¹⁷.0⁵,¹⁴.0⁸,¹³]tetracosan-10-yl acetate |
|---|
| Traditional Name | (1S,4S,5R,10S,13R,17S,18R,19S,20R)-4,5,9,9,13,19,20-heptamethyl-23-oxo-24-oxahexacyclo[15.5.2.0¹,¹⁸.0⁴,¹⁷.0⁵,¹⁴.0⁸,¹³]tetracosan-10-yl acetate |
|---|
| SMILES | [H][C@@]12[C@@H](C)[C@H](C)CC[C@]11CC[C@]3(C)[C@@]2(CCC2[C@@]4(C)CC[C@H](OC(C)=O)C(C)(C)C4CC[C@@]32C)OC1=O |
|---|
| InChI Identifier | InChI=1S/C32H50O4/c1-19-9-15-31-18-17-30(8)29(7)14-10-22-27(4,5)24(35-21(3)33)12-13-28(22,6)23(29)11-16-32(30,36-26(31)34)25(31)20(19)2/h19-20,22-25H,9-18H2,1-8H3/t19-,20+,22?,23?,24+,25-,28+,29-,30+,31+,32+/m1/s1 |
|---|
| InChI Key | NJLCBLDWBHWOFU-KEEAFDKNSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as triterpenoids. These are terpene molecules containing six isoprene units. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Prenol lipids |
|---|
| Sub Class | Triterpenoids |
|---|
| Direct Parent | Triterpenoids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Triterpenoid
- Caprolactone
- Oxepane
- Dicarboxylic acid or derivatives
- Gamma butyrolactone
- Tetrahydrofuran
- Carboxylic acid ester
- Lactone
- Carboxylic acid derivative
- Oxacycle
- Organoheterocyclic compound
- Hydrocarbon derivative
- Organic oxide
- Organic oxygen compound
- Carbonyl group
- Organooxygen compound
- Aliphatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aliphatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-001i-2053900000-39e0148d358df3a9e4a5 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0002-0000900000-ee4958f41f1489583119 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a4s-1014900000-7d0a77e09229c99f6eee | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0pc1-5019800000-b7cf32c8f9d903dedea9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0002-0000900000-c92e45db536976d5dcfa | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4i-2000900000-852696bcdef80a380418 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0bt9-4100900000-e19bb4070260ab31d6d7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4j-9000800000-d0f36065b5f36a5d14f7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4i-9000200000-f2425db2466d4577bfda | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-052b-6000900000-519fd590f84d7a364fec | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-000j-0000900000-cccc354900d094d6ee8b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-000b-0342900000-1b5fef21e41e482685c1 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-000b-1971600000-5f798e52c28066042da1 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0036674 |
|---|
| FooDB ID | FDB015604 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 35014191 |
|---|
| ChEBI ID | 169596 |
|---|
| PubChem Compound ID | 131752031 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|