| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 01:49:19 UTC |
|---|
| Update Date | 2016-11-09 01:19:03 UTC |
|---|
| Accession Number | CHEM030325 |
|---|
| Identification |
|---|
| Common Name | Ketopelenolide a |
|---|
| Class | Small Molecule |
|---|
| Description | Ketopelenolide b is found in alcoholic beverages. Ketopelenolide b is from Artemisia absinthium (wormwood). |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
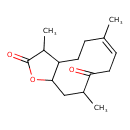
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 3-oxo-1(10)-Germacren-12,6-olide | HMDB | | Ketopelenolid-a | HMDB | | Oxopelenolide a | HMDB |
|
|---|
| Chemical Formula | C15H22O3 |
|---|
| Average Molecular Mass | 250.333 g/mol |
|---|
| Monoisotopic Mass | 250.157 g/mol |
|---|
| CAS Registry Number | 17909-92-1 |
|---|
| IUPAC Name | 3,6,10-trimethyl-2H,3H,3aH,4H,5H,8H,9H,10H,11H,11aH-cyclodeca[b]furan-2,9-dione |
|---|
| Traditional Name | 3,6,10-trimethyl-3H,3aH,4H,5H,8H,10H,11H,11aH-cyclodeca[b]furan-2,9-dione |
|---|
| SMILES | CC1C2CC\C(C)=C/CC(=O)C(C)CC2OC1=O |
|---|
| InChI Identifier | InChI=1S/C15H22O3/c1-9-4-6-12-11(3)15(17)18-14(12)8-10(2)13(16)7-5-9/h5,10-12,14H,4,6-8H2,1-3H3/b9-5- |
|---|
| InChI Key | KLZWSNKEPLKAOS-UITAMQMPSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as germacranolides and derivatives. These are sesquiterpene lactones with a structure based on the germacranolide skeleton, characterized by a gamma lactone fused to a 1,7-dimethylcyclodec-1-ene moiety. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Prenol lipids |
|---|
| Sub Class | Terpene lactones |
|---|
| Direct Parent | Germacranolides and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Germacranolide
- Germacrane sesquiterpenoid
- Sesquiterpenoid
- Gamma butyrolactone
- Tetrahydrofuran
- Carboxylic acid ester
- Ketone
- Lactone
- Cyclic ketone
- Monocarboxylic acid or derivatives
- Carboxylic acid derivative
- Oxacycle
- Organoheterocyclic compound
- Carbonyl group
- Hydrocarbon derivative
- Organic oxide
- Organic oxygen compound
- Organooxygen compound
- Aliphatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aliphatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-004i-7940000000-bbda2c5a6b8c6e8c9c77 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0udi-0290000000-a0a03e0506e0ea85dd31 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0udi-3890000000-5fe492b4d6aabb5dd633 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-1000-9500000000-6f037a2dac50170a67fb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0002-0090000000-e34b4fe85e6fd851bbca | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-052b-0190000000-cff3accc943516cb1e1a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0006-9210000000-28463701c7a16359d049 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0002-0090000000-0dd1e5565d33dd37f20a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-056s-0590000000-9378de90e875d461e723 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-00pi-2790000000-b01858e15d19eb379baf | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0udi-0090000000-a2e6b66e384510551893 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0kei-0690000000-cc5a8f2f186f000c8a54 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-056r-0910000000-08bcb1635047109c1dfc | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0036664 |
|---|
| FooDB ID | FDB015591 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00012210 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 35014186 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 12304831 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|