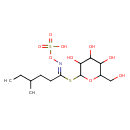| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 01:38:51 UTC |
|---|
| Update Date | 2016-11-09 01:19:00 UTC |
|---|
| Accession Number | CHEM030090 |
|---|
| Identification |
|---|
| Common Name | 3-Methylpentyl glucosinolate |
|---|
| Class | Small Molecule |
|---|
| Description | 3-Methylpentyl glucosinolate is found in brassicas. 3-Methylpentyl glucosinolate is a constituent of Raphanus sativus (radish) and Wasabia japonica (Japanese horseradish) |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 3-Methylpentyl glucosinolic acid | Generator | | 1-thio-b-D-Glucopyranose 1-[4-methyl-N-(sulfooxy)hexanimidate] | HMDB | | {[(e)-(4-methyl-1-{[3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]sulfanyl}hexylidene)amino]oxy}sulfonate | Generator | | {[(e)-(4-methyl-1-{[3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]sulphanyl}hexylidene)amino]oxy}sulphonate | Generator | | {[(e)-(4-methyl-1-{[3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]sulphanyl}hexylidene)amino]oxy}sulphonic acid | Generator |
|
|---|
| Chemical Formula | C13H25NO9S2 |
|---|
| Average Molecular Mass | 403.469 g/mol |
|---|
| Monoisotopic Mass | 403.097 g/mol |
|---|
| CAS Registry Number | Not Available |
|---|
| IUPAC Name | {[(E)-(4-methyl-1-{[3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]sulfanyl}hexylidene)amino]oxy}sulfonic acid |
|---|
| Traditional Name | [(E)-(4-methyl-1-{[3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]sulfanyl}hexylidene)amino]oxysulfonic acid |
|---|
| SMILES | CCC(C)CC\C(SC1OC(CO)C(O)C(O)C1O)=N/OS(O)(=O)=O |
|---|
| InChI Identifier | InChI=1S/C13H25NO9S2/c1-3-7(2)4-5-9(14-23-25(19,20)21)24-13-12(18)11(17)10(16)8(6-15)22-13/h7-8,10-13,15-18H,3-6H2,1-2H3,(H,19,20,21)/b14-9+ |
|---|
| InChI Key | ISQHSPNQOCVWOY-NTEUORMPSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as alkylglucosinolates. These are organic compounds containing a glucosinolate moiety that carries an alkyl chain. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organic oxygen compounds |
|---|
| Class | Organooxygen compounds |
|---|
| Sub Class | Carbohydrates and carbohydrate conjugates |
|---|
| Direct Parent | Alkylglucosinolates |
|---|
| Alternative Parents | |
|---|
| Substituents | - Alkylglucosinolate
- Glycosyl compound
- S-glycosyl compound
- Oxane
- Monothioacetal
- Organic sulfuric acid or derivatives
- Secondary alcohol
- Polyol
- Oxacycle
- Organoheterocyclic compound
- Sulfenyl compound
- Alcohol
- Hydrocarbon derivative
- Organosulfur compound
- Organonitrogen compound
- Organic oxide
- Primary alcohol
- Organic nitrogen compound
- Organopnictogen compound
- Aliphatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aliphatic heteromonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0fe0-9416000000-682b8b0ed6a4a4ef6475 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (4 TMS) - 70eV, Positive | splash10-004i-5011009000-4197d01058fd79c56bd9 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0f7d-6987500000-333bdb0aa832f56c4c4b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-01vp-3690000000-cef9ca6bf5f981d5d6d5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-052r-9000000000-f79a3050314578683e30 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0006-3290000000-5db390e3cad9e84deddc | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4i-8970000000-8eef84bfc2c80a8417c6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0c0r-6930000000-807a9d78a402cfcbd07e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-0210900000-57729e014ac7a6a42dd0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0006-3490100000-eb3ec40f2e10124f0d92 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-01pk-9820000000-b1330d75018d18e141b4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0udi-0001900000-9a124fbe3d77f1327b47 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-114u-1639400000-367feafabb4973a62f56 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-015c-3490000000-4d53203263500a941966 | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0036355 |
|---|
| FooDB ID | FDB015229 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00007827 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 35014122 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 131751970 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|