| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 01:38:34 UTC |
|---|
| Update Date | 2016-11-09 01:19:00 UTC |
|---|
| Accession Number | CHEM030082 |
|---|
| Identification |
|---|
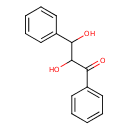
| Common Name | 2,3-Dihydroxy-1,3-diphenyl-1-propanone |
|---|
| Class | Small Molecule |
|---|
| Description | 2,3-Dihydroxy-1,3-diphenyl-1-propanone is found in fats and oils. 2,3-Dihydroxy-1,3-diphenyl-1-propanone is a constituent of peanut oil |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 1-Benzoyl-2-phenyl-1,2-ethanediol | HMDB | | a,b-Dihydroxydihydrochalcone | HMDB |
|
|---|
| Chemical Formula | C15H14O3 |
|---|
| Average Molecular Mass | 242.270 g/mol |
|---|
| Monoisotopic Mass | 242.094 g/mol |
|---|
| CAS Registry Number | Not Available |
|---|
| IUPAC Name | 2,3-dihydroxy-1,3-diphenylpropan-1-one |
|---|
| Traditional Name | 2,3-dihydroxy-1,3-diphenylpropan-1-one |
|---|
| SMILES | OC(C(O)C1=CC=CC=C1)C(=O)C1=CC=CC=C1 |
|---|
| InChI Identifier | InChI=1S/C15H14O3/c16-13(11-7-3-1-4-8-11)15(18)14(17)12-9-5-2-6-10-12/h1-10,13,15-16,18H |
|---|
| InChI Key | QOFVVEZPQRISRL-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as retro-dihydrochalcones. These are a form of normal dihydrochalcones that are structurally distinguished by the lack of oxygen functionalities at the C2'- and C6'-positions. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Phenylpropanoids and polyketides |
|---|
| Class | Linear 1,3-diarylpropanoids |
|---|
| Sub Class | Chalcones and dihydrochalcones |
|---|
| Direct Parent | Retro-dihydrochalcones |
|---|
| Alternative Parents | |
|---|
| Substituents | - Retro-dihydrochalcone
- Alkyl-phenylketone
- Butyrophenone
- Phenylketone
- Benzoyl
- Aryl alkyl ketone
- Aryl ketone
- Acyloin
- Monocyclic benzene moiety
- Beta-hydroxy ketone
- Monosaccharide
- Benzenoid
- Alpha-hydroxy ketone
- Ketone
- 1,2-diol
- Secondary alcohol
- Organic oxide
- Alcohol
- Organic oxygen compound
- Organooxygen compound
- Aromatic alcohol
- Hydrocarbon derivative
- Aromatic homomonocyclic compound
|
|---|
| Molecular Framework | Aromatic homomonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0a4i-4900000000-0af49e44025168127216 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (2 TMS) - 70eV, Positive | splash10-056r-2900000000-b81076630d2284d497f6 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0006-0390000000-309e3e1a654b66869124 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a4i-1910000000-e3cb137fd80e52eeb5dd | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a4i-9800000000-892e3c90f8b9bb5aa351 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0006-0390000000-a54889f4b772d4fe5cf3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-056r-7920000000-55dda432a8b1f48bbc17 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-004i-9300000000-f7ab78f19929290b9044 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a6u-0390000000-64f3687072acc6050e8e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a6r-4590000000-a39c62661d424990c236 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0006-9100000000-247fd27b69a86f78945c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0076-0790000000-bab6ecf34e5ce7cf008c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a59-2920000000-3be6ae16de90c3a42f08 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-00or-6900000000-7901cdfd8b339c84fa3a | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0036347 |
|---|
| FooDB ID | FDB015218 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 4475891 |
|---|
| ChEBI ID | 173745 |
|---|
| PubChem Compound ID | 5316933 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|