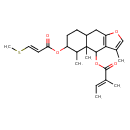Identification Common Name S-Furanopetasitin Class Small Molecule Description S-Furanopetasitin is found in giant butterbur. S-Furanopetasitin is a constituent of Petasites japonicus (sweet coltsfoot) Contaminant Sources Contaminant Type Not Available Chemical Structure Synonyms Value Source (S)-Furanopetasitin HMDB 3,4a,5-Trimethyl-6-{[(2E)-3-(methylsulfanyl)prop-2-enoyl]oxy}-4H,4ah,5H,6H,7H,8H,8ah,9H-naphtho[2,3-b]furan-4-yl (2E)-2-methylbut-2-enoic acid Generator 3,4a,5-Trimethyl-6-{[(2E)-3-(methylsulphanyl)prop-2-enoyl]oxy}-4H,4ah,5H,6H,7H,8H,8ah,9H-naphtho[2,3-b]furan-4-yl (2E)-2-methylbut-2-enoate Generator 3,4a,5-Trimethyl-6-{[(2E)-3-(methylsulphanyl)prop-2-enoyl]oxy}-4H,4ah,5H,6H,7H,8H,8ah,9H-naphtho[2,3-b]furan-4-yl (2E)-2-methylbut-2-enoic acid Generator
Chemical Formula C24 H32 O5 S Average Molecular Mass 432.573 g/mol Monoisotopic Mass 432.197 g/mol CAS Registry Number 34335-97-2 IUPAC Name 3,4a,5-trimethyl-6-{[(2E)-3-(methylsulfanyl)prop-2-enoyl]oxy}-4H,4aH,5H,6H,7H,8H,8aH,9H-naphtho[2,3-b]furan-4-yl (2E)-2-methylbut-2-enoate Traditional Name 3,4a,5-trimethyl-6-{[(2E)-3-(methylsulfanyl)prop-2-enoyl]oxy}-4H,5H,6H,7H,8H,8aH,9H-naphtho[2,3-b]furan-4-yl (2E)-2-methylbut-2-enoate SMILES CS\C=C\C(=O)OC1CCC2CC3=C(C(OC(=O)C(\C)=C\C)C2(C)C1C)C(C)=CO3 InChI Identifier InChI=1S/C24H32O5S/c1-7-14(2)23(26)29-22-21-15(3)13-27-19(21)12-17-8-9-18(16(4)24(17,22)5)28-20(25)10-11-30-6/h7,10-11,13,16-18,22H,8-9,12H2,1-6H3/b11-10+,14-7+ InChI Key DZIJJEZRPMYRRP-QFPWAJRASA-N Chemical Taxonomy Description belongs to the class of organic compounds known as eremophilane, 8,9-secoeremophilane and furoeremophilane sesquiterpenoids. These are sesquiterpenoids with a structure based either on the eremophilane skeleton, its 8,9-seco derivative, or the furoeremophilane skeleton. Eremophilanes have been shown to be derived from eudesmanes by migration of the methyl group at C-10 to C-5. Kingdom Organic compounds Super Class Lipids and lipid-like molecules Class Prenol lipids Sub Class Sesquiterpenoids Direct Parent Eremophilane, 8,9-secoeremophilane and furoeremophilane sesquiterpenoids Alternative Parents Substituents Furoeremophilane sesquiterpenoid
Naphthofuran
Benzofuran
Fatty acid ester
Dicarboxylic acid or derivatives
Fatty acyl
Vinylogous thioester
Heteroaromatic compound
Acrylic acid or derivatives
Furan
Alpha,beta-unsaturated carboxylic ester
Enoate ester
Carboxylic acid ester
Thioenolether
Organoheterocyclic compound
Carboxylic acid derivative
Oxacycle
Sulfenyl compound
Organosulfur compound
Organooxygen compound
Organic oxide
Hydrocarbon derivative
Organic oxygen compound
Carbonyl group
Aromatic heteropolycyclic compound Molecular Framework Aromatic heteropolycyclic compounds External Descriptors Not Available