| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 01:31:23 UTC |
|---|
| Update Date | 2016-11-09 01:18:58 UTC |
|---|
| Accession Number | CHEM029929 |
|---|
| Identification |
|---|
| Common Name | Dehydro-1,8-cineole |
|---|
| Class | Small Molecule |
|---|
| Description | Dehydro-1,8-cineole is found in herbs and spices. Dehydro-1,8-cineole is isolated from Laurus nobilis (bay laurel) oi |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
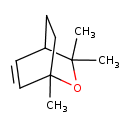
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 1,3,3-Trimethyl-2-oxabicyclo[2.2.2]oct-5-ene | HMDB | | 1,8-Cineole dehydro | HMDB | | 1,8-Dehydrocineole | HMDB | | 1,8-Epoxy-P-menth-2-ene | HMDB | | 2,3-dehydro-1,8-Cineol | HMDB | | 2,3-dehydro-1,8-Cineole | HMDB | | dehydro-1,8-Cineol | HMDB | | dehydro-1,8-Cyneole | HMDB | | dehydro-1.8-Cineole | HMDB | | Dehydrocineole | HMDB |
|
|---|
| Chemical Formula | C10H16O |
|---|
| Average Molecular Mass | 152.233 g/mol |
|---|
| Monoisotopic Mass | 152.120 g/mol |
|---|
| CAS Registry Number | 92760-25-3 |
|---|
| IUPAC Name | 1,3,3-trimethyl-2-oxabicyclo[2.2.2]oct-5-ene |
|---|
| Traditional Name | 1,3,3-trimethyl-2-oxabicyclo[2.2.2]oct-5-ene |
|---|
| SMILES | CC1(C)OC2(C)CCC1C=C2 |
|---|
| InChI Identifier | InChI=1S/C10H16O/c1-9(2)8-4-6-10(3,11-9)7-5-8/h4,6,8H,5,7H2,1-3H3 |
|---|
| InChI Key | LOOYOTLEOHYYOV-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as pyrans. Pyrans are compounds containing a pyran ring, which is a six-member heterocyclic, non-aromatic ring with five carbon atoms, one oxygen atom and two ring double bonds. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Pyrans |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Pyrans |
|---|
| Alternative Parents | |
|---|
| Substituents | - Pyran
- Oxane
- Oxacycle
- Ether
- Dialkyl ether
- Organic oxygen compound
- Hydrocarbon derivative
- Organooxygen compound
- Aliphatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aliphatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-000i-2900000000-5510b29ed704dc2501b9 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0udi-0900000000-e3088a8b47e3e53258c9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0udi-0900000000-3c4f7cd82aae50c9081a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0f79-0900000000-fdf7821c709c68afaad2 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-0900000000-4e47e22648e40cab16c5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0udi-0900000000-4e47e22648e40cab16c5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0f79-0900000000-dc7c90c139b8939e6d4d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0udi-0900000000-0d7f2c31f0ee3340d4f0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0udi-0900000000-0d7f2c31f0ee3340d4f0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0udi-0900000000-5655efdcc74fbd2afcfc | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-0900000000-c373c9eea3cebf186f53 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0udi-0900000000-c373c9eea3cebf186f53 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0udi-0900000000-619f4705f851bbd9f793 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0036085 |
|---|
| FooDB ID | FDB014921 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00055792 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 456244 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 523035 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|