| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 01:27:57 UTC |
|---|
| Update Date | 2016-11-09 01:18:57 UTC |
|---|
| Accession Number | CHEM029853 |
|---|
| Identification |
|---|
| Common Name | Ganoderic acid Mg |
|---|
| Class | Small Molecule |
|---|
| Description | Ganoderic acid Mg is found in mushrooms. Ganoderic acid Mg is a metabolite Ganoderma lucidum (reishi |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
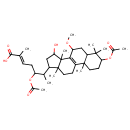
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Ganoderate MG | Generator | | 3a,22-Diacetoxy-15a-hydroxy-7a-methoxylanosta-8,24E-dien-26-Oic acid | HMDB | | 3alpha,22-Diacetoxy-15alpha-hydroxy-7alpha-methoxy-5alpha-lanosta-8,24E-dien-26-Oic acid | HMDB | | (2E)-5-(Acetyloxy)-6-[5-(acetyloxy)-12-hydroxy-9-methoxy-2,6,6,11,15-pentamethyltetracyclo[8.7.0.0²,⁷.0¹¹,¹⁵]heptadec-1(10)-en-14-yl]-2-methylhept-2-enoate | Generator |
|
|---|
| Chemical Formula | C35H54O8 |
|---|
| Average Molecular Mass | 602.799 g/mol |
|---|
| Monoisotopic Mass | 602.382 g/mol |
|---|
| CAS Registry Number | 110042-11-0 |
|---|
| IUPAC Name | (2E)-5-(acetyloxy)-6-[5-(acetyloxy)-12-hydroxy-9-methoxy-2,6,6,11,15-pentamethyltetracyclo[8.7.0.0²,⁷.0¹¹,¹⁵]heptadec-1(10)-en-14-yl]-2-methylhept-2-enoic acid |
|---|
| Traditional Name | (2E)-5-(acetyloxy)-6-[5-(acetyloxy)-12-hydroxy-9-methoxy-2,6,6,11,15-pentamethyltetracyclo[8.7.0.0²,⁷.0¹¹,¹⁵]heptadec-1(10)-en-14-yl]-2-methylhept-2-enoic acid |
|---|
| SMILES | COC1CC2C(C)(C)C(CCC2(C)C2=C1C1(C)C(O)CC(C(C)C(C\C=C(/C)C(O)=O)OC(C)=O)C1(C)CC2)OC(C)=O |
|---|
| InChI Identifier | InChI=1S/C35H54O8/c1-19(31(39)40)11-12-25(42-21(3)36)20(2)24-17-28(38)35(9)30-23(13-16-34(24,35)8)33(7)15-14-29(43-22(4)37)32(5,6)27(33)18-26(30)41-10/h11,20,24-29,38H,12-18H2,1-10H3,(H,39,40)/b19-11+ |
|---|
| InChI Key | SSISLAMFCZQHEH-YBFXNURJSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as triterpenoids. These are terpene molecules containing six isoprene units. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Prenol lipids |
|---|
| Sub Class | Triterpenoids |
|---|
| Direct Parent | Triterpenoids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Triterpenoid
- Hydroxy bile acid, alcohol, or derivatives
- Monohydroxy bile acid, alcohol, or derivatives
- Bile acid, alcohol, or derivatives
- Steroid ester
- Steroid acid
- 15-hydroxysteroid
- Hydroxysteroid
- Steroid
- Tricarboxylic acid or derivatives
- Cyclic alcohol
- Secondary alcohol
- Carboxylic acid ester
- Ether
- Dialkyl ether
- Carboxylic acid
- Carboxylic acid derivative
- Alcohol
- Hydrocarbon derivative
- Organic oxide
- Carbonyl group
- Organic oxygen compound
- Organooxygen compound
- Aliphatic homopolycyclic compound
|
|---|
| Molecular Framework | Aliphatic homopolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0f6x-1001390000-6a5e5cd5b08067bfe700 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (1 TMS) - 70eV, Positive | splash10-0cmi-3000339000-f36e629722e1fcca82d1 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_2) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_1) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_1_1) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_1_2) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_2_1) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS ("Ganoderic acid Mg,1TMS,#1" TMS) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0gw3-0000091000-d652a133c85529c89b26 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-01rf-0000190000-1c401d0397d7985c8874 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-03xs-0100690000-2cf6a2739435c4b07b1f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0zfu-1000094000-f41a9412e9ec6f7d50c2 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4l-1000090000-87086edbadcb18013e4d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-05mo-4000190000-5112d55a61109d6aa428 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-001i-0200930000-7cf771fe22cf0b7f10d3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-01tj-0500690000-bdafbf16ce0d80700990 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0ik9-4901000000-88e8eb32dbea87e62b5a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0a4i-9000101000-59f8d0035a5fa82c2652 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0a4i-9000143000-ed365765759267321676 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a6r-9700010000-a4e63aad65e7ea259c39 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0035999 |
|---|
| FooDB ID | FDB014808 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00023838 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 74886445 |
|---|
| ChEBI ID | 191487 |
|---|
| PubChem Compound ID | 131751896 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|