| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 01:21:14 UTC |
|---|
| Update Date | 2016-11-09 01:18:56 UTC |
|---|
| Accession Number | CHEM029704 |
|---|
| Identification |
|---|
| Common Name | Aubergenone |
|---|
| Class | Small Molecule |
|---|
| Description | Aubergenone is found in eggplant. Aubergenone is found in diseased eggplant (Solanum melongena) and from the stem bark of Mangifera indica (mango |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
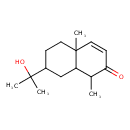
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Mangeudesmenone | HMDB | | [1R-(1alpha,4Aalpha,7alpha,8abeta)]-4a,5,6,7,8,8a-hexahydro-7-(1-hydroxy-1-methylethyl)-1,4a-dimethyl-2(1H)-naphthalenone | HMDB |
|
|---|
| Chemical Formula | C15H24O2 |
|---|
| Average Molecular Mass | 236.350 g/mol |
|---|
| Monoisotopic Mass | 236.178 g/mol |
|---|
| CAS Registry Number | 69427-35-6 |
|---|
| IUPAC Name | 7-(2-hydroxypropan-2-yl)-1,4a-dimethyl-1,2,4a,5,6,7,8,8a-octahydronaphthalen-2-one |
|---|
| Traditional Name | 7-(2-hydroxypropan-2-yl)-1,4a-dimethyl-1,5,6,7,8,8a-hexahydronaphthalen-2-one |
|---|
| SMILES | CC1C2CC(CCC2(C)C=CC1=O)C(C)(C)O |
|---|
| InChI Identifier | InChI=1S/C15H24O2/c1-10-12-9-11(14(2,3)17)5-7-15(12,4)8-6-13(10)16/h6,8,10-12,17H,5,7,9H2,1-4H3 |
|---|
| InChI Key | MSSWKGZMPUXALD-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as eudesmane, isoeudesmane or cycloeudesmane sesquiterpenoids. These are sesquiterpenoids with a structure based on the eudesmane skeleton. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Prenol lipids |
|---|
| Sub Class | Sesquiterpenoids |
|---|
| Direct Parent | Eudesmane, isoeudesmane or cycloeudesmane sesquiterpenoids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Eudesmane, isoeudesmane or cycloeudesmane sesquiterpenoid
- Cyclohexenone
- Tertiary alcohol
- Cyclic ketone
- Ketone
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Carbonyl group
- Alcohol
- Aliphatic homopolycyclic compound
|
|---|
| Molecular Framework | Aliphatic homopolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0ab9-3950000000-f6a32864344582bb94e4 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (1 TMS) - 70eV, Positive | splash10-003l-5590000000-392d36b59728e6f9ed3d | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014r-0290000000-03816e1c3e313355b05e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00p0-1940000000-817dcda0076697396ca5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0gc1-9710000000-c4b96a33197de6a924cf | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-000i-0090000000-7586dd0d73ed89677ad2 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-002r-0390000000-022a7378257ac0859d48 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-02vi-2960000000-879779dbdcb4ca6510f3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0gbi-0390000000-87fe8281248f42496d53 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-000i-3930000000-72b9a28b8aebb761a4fd | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-008c-9400000000-cf776e3f7223796666fb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-000i-0090000000-c0031dc201eac6b6350a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-000i-0090000000-429531043a4aef925aee | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-067r-0290000000-9685bb5bcc173b218df9 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0035827 |
|---|
| FooDB ID | FDB014591 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00012854 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 35014028 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 15601417 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|