| Identification |
|---|
| Common Name | Ziziphin |
|---|
| Class | Small Molecule |
|---|
| Description | Ziziphin is found in fruits. Ziziphin is a constituent of leaves of Zizyphus jujuba (Chinese date) |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
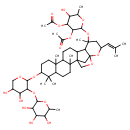
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Zizyphin | HMDB | | 4-(Acetyloxy)-2-{[7-({4,5-dihydroxy-3-[(3,4,5-trihydroxy-6-methyloxan-2-yl)oxy]oxan-2-yl}oxy)-2,6,6,10,16-pentamethyl-18-(2-methylprop-1-en-1-yl)-19,21-dioxahexacyclo[18.2.1.0¹,¹⁴.0²,¹¹.0⁵,¹⁰.0¹⁵,²⁰]tricosan-16-yl]oxy}-5-hydroxy-6-methyloxan-3-yl acetic acid | Generator |
|
|---|
| Chemical Formula | C51H80O18 |
|---|
| Average Molecular Mass | 981.170 g/mol |
|---|
| Monoisotopic Mass | 980.534 g/mol |
|---|
| CAS Registry Number | 73667-51-3 |
|---|
| IUPAC Name | 3-(acetyloxy)-2-{[7-({4,5-dihydroxy-3-[(3,4,5-trihydroxy-6-methyloxan-2-yl)oxy]oxan-2-yl}oxy)-2,6,6,10,16-pentamethyl-18-(2-methylprop-1-en-1-yl)-19,21-dioxahexacyclo[18.2.1.0¹,¹⁴.0²,¹¹.0⁵,¹⁰.0¹⁵,²⁰]tricosan-16-yl]oxy}-5-hydroxy-6-methyloxan-4-yl acetate |
|---|
| Traditional Name | 3-(acetyloxy)-2-{[7-({4,5-dihydroxy-3-[(3,4,5-trihydroxy-6-methyloxan-2-yl)oxy]oxan-2-yl}oxy)-2,6,6,10,16-pentamethyl-18-(2-methylprop-1-en-1-yl)-19,21-dioxahexacyclo[18.2.1.0¹,¹⁴.0²,¹¹.0⁵,¹⁰.0¹⁵,²⁰]tricosan-16-yl]oxy}-5-hydroxy-6-methyloxan-4-yl acetate |
|---|
| SMILES | CC1OC(OC2C(O)C(O)COC2OC2CCC3(C)C(CCC4(C)C3CCC3C5C6(CC43CO6)OC(CC5(C)OC3OC(C)C(O)C(OC(C)=O)C3OC(C)=O)C=C(C)C)C2(C)C)C(O)C(O)C1O |
|---|
| InChI Identifier | InChI=1S/C51H80O18/c1-23(2)18-28-19-49(11,69-45-41(65-27(6)53)39(64-26(5)52)35(56)25(4)63-45)42-29-12-13-32-47(9)16-15-33(46(7,8)31(47)14-17-48(32,10)50(29)21-51(42,68-28)61-22-50)66-44-40(36(57)30(54)20-60-44)67-43-38(59)37(58)34(55)24(3)62-43/h18,24-25,28-45,54-59H,12-17,19-22H2,1-11H3 |
|---|
| InChI Key | MHCWKKSYGWYURT-UHFFFAOYSA-N |
|---|