| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 01:03:46 UTC |
|---|
| Update Date | 2016-11-09 01:18:51 UTC |
|---|
| Accession Number | CHEM029281 |
|---|
| Identification |
|---|
| Common Name | Licoagrodin |
|---|
| Class | Small Molecule |
|---|
| Description | Isolated from hairy root cultures of Glycyrrhiza glabra (licorice). Licoagrodin is found in tea and herbs and spices. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
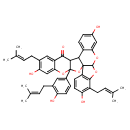
| Chemical Structure | |
|---|
| Synonyms | Not Available |
|---|
| Chemical Formula | C45H44O9 |
|---|
| Average Molecular Mass | 728.826 g/mol |
|---|
| Monoisotopic Mass | 728.299 g/mol |
|---|
| CAS Registry Number | Not Available |
|---|
| IUPAC Name | 5,13,23-trihydroxy-27-[4-hydroxy-3-(3-methylbut-2-en-1-yl)phenyl]-6,22-bis(3-methylbut-2-en-1-yl)-8,10,26,28-tetraoxaheptacyclo[15.11.0.0¹,⁹.0²,⁷.0¹¹,¹⁶.0¹⁸,²⁷.0²⁰,²⁵]octacosa-2(7),3,5,11,13,15,20,22,24-nonaen-19-one |
|---|
| Traditional Name | 5,13,23-trihydroxy-27-[4-hydroxy-3-(3-methylbut-2-en-1-yl)phenyl]-6,22-bis(3-methylbut-2-en-1-yl)-8,10,26,28-tetraoxaheptacyclo[15.11.0.0¹,⁹.0²,⁷.0¹¹,¹⁶.0¹⁸,²⁷.0²⁰,²⁵]octacosa-2(7),3,5,11,13,15,20,22,24-nonaen-19-one |
|---|
| SMILES | CC(C)=CCC1=C(O)C=CC(=C1)C12OC34C(OC5=C3C=CC(O)=C5CC=C(C)C)OC3=CC(O)=CC=C3C4C1C(=O)C1=CC(CC=C(C)C)=C(O)C=C1O2 |
|---|
| InChI Identifier | InChI=1S/C45H44O9/c1-23(2)7-10-26-19-28(12-17-34(26)47)45-40(41(50)32-20-27(11-8-24(3)4)36(49)22-38(32)53-45)39-31-15-13-29(46)21-37(31)51-43-44(39,54-45)33-16-18-35(48)30(42(33)52-43)14-9-25(5)6/h7-9,12-13,15-22,39-40,43,46-49H,10-11,14H2,1-6H3 |
|---|
| InChI Key | IUNNUKJWKPJUCQ-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as 6-prenylated flavanones. These are flavanones that features a C5-isoprenoid substituent at the 6-position. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Phenylpropanoids and polyketides |
|---|
| Class | Flavonoids |
|---|
| Sub Class | Flavans |
|---|
| Direct Parent | 6-prenylated flavanones |
|---|
| Alternative Parents | |
|---|
| Substituents | - 3'-prenylated flavanone
- 6-prenylated flavanone
- 3'-prenylated flavan
- 4'-hydroxyflavonoid
- 7-hydroxyflavonoid
- Hydroxyflavonoid
- Coumaronochromene
- Isoflavonoid
- Isoflavan
- Chromone
- Chromane
- Benzopyran
- 1-benzopyran
- Coumaran
- Furopyran
- Aryl alkyl ketone
- Aryl ketone
- Phenol
- Ketal
- 1-hydroxy-2-unsubstituted benzenoid
- Monocyclic benzene moiety
- Benzenoid
- Pyran
- Tetrahydrofuran
- Furan
- Ketone
- Acetal
- Organoheterocyclic compound
- Oxacycle
- Polyol
- Organic oxide
- Hydrocarbon derivative
- Organic oxygen compound
- Organooxygen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_1) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_2) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_3) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_1_4) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_1) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_2) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_3) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_4) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_5) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TMS_2_6) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_1_1) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_1_2) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_1_3) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (TBDMS_1_4) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-002r-0314019800-bdaac93c0f5f0b099314 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-02mr-1210009200-de6b14ef9eefb054769a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0avi-2900011000-2c00719720046dd05004 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-0300000900-65770c1f641405e4730e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0adi-0203073900-a1b2fcaaefaf645ef103 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-0913110100-b01c32ad1f315f46dbc1 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-0000000900-165983fa8148d80fb566 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0fb9-0070000900-3e8ccfdf1c8dbd4ed916 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-00di-0010090000-f762a6756b80027f6383 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-004i-0000000900-43e9d5b12bd3670e4b65 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a7i-0090400600-c295fff06780799debbe | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a4i-0090120000-3506b763b0fdc5be7c00 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0035372 |
|---|
| FooDB ID | FDB014046 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00014716 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 78200726 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|