| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 01:03:31 UTC |
|---|
| Update Date | 2016-11-09 01:18:51 UTC |
|---|
| Accession Number | CHEM029275 |
|---|
| Identification |
|---|
| Common Name | Cytochalasin Opho |
|---|
| Class | Small Molecule |
|---|
| Description | Cytochalasin Opho is a mycotoxin produced by Phomopsis sp |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
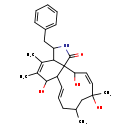
| Chemical Structure | |
|---|
| Synonyms | Not Available |
|---|
| Chemical Formula | C28H37NO4 |
|---|
| Average Molecular Mass | 451.598 g/mol |
|---|
| Monoisotopic Mass | 451.272 g/mol |
|---|
| CAS Registry Number | 108050-26-6 |
|---|
| IUPAC Name | 3-benzyl-6,12,15-trihydroxy-4,5,10,12-tetramethyl-1H,2H,3H,6H,6aH,9H,10H,11H,12H,15H,15bH-cycloundeca[e]isoindol-1-one |
|---|
| Traditional Name | 3-benzyl-6,12,15-trihydroxy-4,5,10,12-tetramethyl-2H,3H,6H,6aH,9H,10H,11H,15H,15bH-cycloundeca[e]isoindol-1-one |
|---|
| SMILES | CC1C\C=C\C2C(O)C(C)=C(C)C3C(CC4=CC=CC=C4)NC(=O)C23C(O)\C=C/C(C)(O)C1 |
|---|
| InChI Identifier | InChI=1S/C28H37NO4/c1-17-9-8-12-21-25(31)19(3)18(2)24-22(15-20-10-6-5-7-11-20)29-26(32)28(21,24)23(30)13-14-27(4,33)16-17/h5-8,10-14,17,21-25,30-31,33H,9,15-16H2,1-4H3,(H,29,32)/b12-8+,14-13- |
|---|
| InChI Key | UMHVFKLUODBPSC-JUDANRDHSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as cytochalasans. These are fungal metabolites structurally characterized by the presence of an isoindolone nucleus fused to a macrocyclic ring, which can either a lactone, as in cytochalasin B, a carbonate, as in cytochalasin E, or a carbocycle, as in cytochalasin D, H, and K. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Alkaloids and derivatives |
|---|
| Class | Cytochalasans |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Cytochalasans |
|---|
| Alternative Parents | |
|---|
| Substituents | - Carbocyclic cytochalasan skeleton
- Cytochalasan
- Isoindolone
- Isoindoline
- Isoindole
- Isoindole or derivatives
- Monocyclic benzene moiety
- Pyrrolidone
- 2-pyrrolidone
- Benzenoid
- Tertiary alcohol
- Pyrrolidine
- Secondary alcohol
- Secondary carboxylic acid amide
- Carboxamide group
- Lactam
- Organoheterocyclic compound
- Polyol
- Carboxylic acid derivative
- Azacycle
- Organic oxide
- Organopnictogen compound
- Organic oxygen compound
- Organic nitrogen compound
- Alcohol
- Carbonyl group
- Hydrocarbon derivative
- Organooxygen compound
- Organonitrogen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-001u-6106900000-e8be727ce773a090e1e6 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (3 TMS) - 70eV, Positive | splash10-0ufr-9000058000-fb48ae281c3f38af7573 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00lr-0000900000-0c1c31078df97cdd3bcd | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0159-1003900000-ed9e673f37f49d775344 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0ktf-9803100000-1e05a140d608acd0eb85 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-0000900000-18cd4bde26774045bec6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0f89-0002900000-1212eba7380cc245c7ba | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0006-9023100000-4d564ef3e4d3057de571 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-0000900000-63c7d2587ab33f731cab | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0udi-0000900000-1490130e0cb5aed0f31b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-00kg-4004900000-8e3ece949c94684a155c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0ue9-0000900000-7c0492428ce5b962ce86 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-001i-0001900000-c010c5b441ff3d1244a7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0gi3-2209300000-e2981ba1bba8c6cf65f0 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0035366 |
|---|
| FooDB ID | FDB014039 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00011340 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 35013910 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 131751724 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|