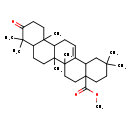
Identification Common Name Methyl 3-oxo-12-oleanen-28-oate Class Small Molecule Description Methyl 3-oxo-12-oleanen-28-oate is found in fruits. Methyl 3-oxo-12-oleanen-28-oate is isolated from Pistacia vera (pistachio) and Zizyphus jujuba (Chinese date). Contaminant Sources Contaminant Type Not Available Chemical Structure Synonyms Value Source Methyl 3-oxo-12-oleanen-28-Oic acid Generator 1-((4-Methylphenyl)sulfonyl)-piperidine HMDB 1-((4-Methylphenyl)sulfonyl)piperidine HMDB 1-(P-Tolylsulfonyl)-piperidine HMDB 1-(P-Tolylsulfonyl)piperidine HMDB 1-(Toluene-4-sulfonyl)-piperidine HMDB 1-Tosylpiperidine HMDB 1-[(4-Methylphenyl)sulfonyl]-piperidine HMDB 1-[(4-Methylphenyl)sulfonyl]piperidine HMDB N-(P-Tolylsulfonyl)piperidine HMDB Methyl 2,2,6a,6b,9,9,12a-heptamethyl-10-oxo-1,2,3,4,4a,5,6,6a,6b,7,8,8a,9,10,11,12,12a,12b,13,14b-icosahydropicene-4a-carboxylic acid Generator
Chemical Formula C31 H48 O3 Average Molecular Mass 468.711 g/mol Monoisotopic Mass 468.360 g/mol CAS Registry Number 1721-58-0 IUPAC Name methyl 2,2,6a,6b,9,9,12a-heptamethyl-10-oxo-1,2,3,4,4a,5,6,6a,6b,7,8,8a,9,10,11,12,12a,12b,13,14b-icosahydropicene-4a-carboxylate Traditional Name methyl 2,2,6a,6b,9,9,12a-heptamethyl-10-oxo-3,4,5,6,7,8,8a,11,12,12b,13,14b-dodecahydro-1H-picene-4a-carboxylate SMILES COC(=O)C12CCC(C)(C)CC1C1=CCC3C4(C)CCC(=O)C(C)(C)C4CCC3(C)C1(C)CC2 InChI Identifier InChI=1S/C31H48O3/c1-26(2)15-17-31(25(33)34-8)18-16-29(6)20(21(31)19-26)9-10-23-28(5)13-12-24(32)27(3,4)22(28)11-14-30(23,29)7/h9,21-23H,10-19H2,1-8H3 InChI Key PPMUFCXCVKVCSV-UHFFFAOYSA-N