| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 00:50:46 UTC |
|---|
| Update Date | 2016-11-09 01:18:48 UTC |
|---|
| Accession Number | CHEM028989 |
|---|
| Identification |
|---|
| Common Name | Gibberellin A13 |
|---|
| Class | Small Molecule |
|---|
| Description | Gibberellin a13 is a member of the class of compounds known as c20-gibberellin 20-carboxylic acids. C20-gibberellin 20-carboxylic acids are c20-gibberellins with a carboxyl group at the 20-position. Gibberellin a13 is slightly soluble (in water) and a weakly acidic compound (based on its pKa). Gibberellin a13 can be found in a number of food items such as japanese pumpkin, catjang pea, boysenberry, and carob, which makes gibberellin a13 a potential biomarker for the consumption of these food products. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
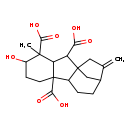
| Chemical Structure | |
|---|
| Synonyms | Not Available |
|---|
| Chemical Formula | C20H26O7 |
|---|
| Average Molecular Mass | 378.416 g/mol |
|---|
| Monoisotopic Mass | 378.168 g/mol |
|---|
| CAS Registry Number | 2922-24-9 |
|---|
| IUPAC Name | 5-hydroxy-4-methyl-13-methylidenetetracyclo[10.2.1.0¹,⁹.0³,⁸]pentadecane-2,4,8-tricarboxylic acid |
|---|
| Traditional Name | 5-hydroxy-4-methyl-13-methylidenetetracyclo[10.2.1.0¹,⁹.0³,⁸]pentadecane-2,4,8-tricarboxylic acid |
|---|
| SMILES | CC1(C2C(C(O)=O)C34CC(CCC3C2(CCC1O)C(O)=O)C(=C)C4)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C20H26O7/c1-9-7-19-8-10(9)3-4-11(19)20(17(26)27)6-5-12(21)18(2,16(24)25)14(20)13(19)15(22)23/h10-14,21H,1,3-8H2,2H3,(H,22,23)(H,24,25)(H,26,27) |
|---|
| InChI Key | UYRCHWLYXIQJKK-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as c20-gibberellin 20-carboxylic acids. These are c20-gibberellins with a carboxyl group at the 6-position. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Prenol lipids |
|---|
| Sub Class | Diterpenoids |
|---|
| Direct Parent | C20-gibberellin 20-carboxylic acids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Gibberellane-20-carboxylic acid
- Gibberellane-6-carboxylic acid
- Tricarboxylic acid or derivatives
- Beta-hydroxy acid
- Hydroxy acid
- Cyclic alcohol
- Secondary alcohol
- Carboxylic acid derivative
- Carboxylic acid
- Organic oxide
- Alcohol
- Carbonyl group
- Organic oxygen compound
- Hydrocarbon derivative
- Organooxygen compound
- Aliphatic homopolycyclic compound
|
|---|
| Molecular Framework | Aliphatic homopolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0403-0009000000-99dda88ab0ec2a227e3b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-02t9-0129000000-c3aebba42c74021930df | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-014r-0396000000-7ce2c83d23fd18ed8e7c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0059-0019000000-785f4cee2d77b660edc9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00li-0049000000-a22e020c600dba187780 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-000i-1094000000-f66c8de991c26e58dc82 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-0009000000-99724dfafe1064c1e28e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-000i-0094000000-cc1a7a6db57e0eec2efb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-004m-9723000000-51e0cf2e1e4d7baf37cb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00or-0009000000-2a961e2293b4d7e60fbc | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00kr-0079000000-bb3fbb1efc3d55f4c392 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0gw0-1791000000-bfb7403713e601a56b5e | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0303455 |
|---|
| FooDB ID | FDB013661 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 24808174 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 12310176 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|