| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 00:50:35 UTC |
|---|
| Update Date | 2016-11-09 01:18:48 UTC |
|---|
| Accession Number | CHEM028985 |
|---|
| Identification |
|---|
| Common Name | Gibberellin A58 |
|---|
| Class | Small Molecule |
|---|
| Description | Alkaloid from stem bark of Murraya koenigii (curryleaf tree). Clausine L is found in herbs and spices. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
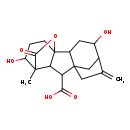
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 2-Methoxy-3-carbomethoxycarbazole | HMDB | | O-Methylmukonidine | HMDB | | GA58 | HMDB | | 4,12-Dihydroxy-11-methyl-6-methylidene-16-oxo-15-oxapentacyclo[9.3.2.1⁵,⁸.0¹,¹⁰.0²,⁸]heptadecane-9-carboxylate | Generator |
|
|---|
| Chemical Formula | C19H24O6 |
|---|
| Average Molecular Mass | 348.390 g/mol |
|---|
| Monoisotopic Mass | 348.157 g/mol |
|---|
| CAS Registry Number | 77288-90-5 |
|---|
| IUPAC Name | 4,12-dihydroxy-11-methyl-6-methylidene-16-oxo-15-oxapentacyclo[9.3.2.1⁵,⁸.0¹,¹⁰.0²,⁸]heptadecane-9-carboxylic acid |
|---|
| Traditional Name | 4,12-dihydroxy-11-methyl-6-methylidene-16-oxo-15-oxapentacyclo[9.3.2.1⁵,⁸.0¹,¹⁰.0²,⁸]heptadecane-9-carboxylic acid |
|---|
| SMILES | CC12C3C(C(O)=O)C45CC(C(O)CC4C3(CCC1O)OC2=O)C(=C)C5 |
|---|
| InChI Identifier | InChI=1S/C19H24O6/c1-8-6-18-7-9(8)10(20)5-11(18)19-4-3-12(21)17(2,16(24)25-19)14(19)13(18)15(22)23/h9-14,20-21H,1,3-7H2,2H3,(H,22,23) |
|---|
| InChI Key | VFOHPKBOIYTDJT-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as carbazoles. Carbazoles are compounds containing a three ring system containing a pyrrole ring fused on either side to a benzene ring. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Indoles and derivatives |
|---|
| Sub Class | Carbazoles |
|---|
| Direct Parent | Carbazoles |
|---|
| Alternative Parents | |
|---|
| Substituents | - Carbazole
- Indolecarboxylic acid
- Indolecarboxylic acid derivative
- O-methoxybenzoic acid or derivatives
- Indole
- Anisole
- Alkyl aryl ether
- Benzenoid
- Heteroaromatic compound
- Methyl ester
- Pyrrole
- Carboxylic acid ester
- Carboxylic acid derivative
- Ether
- Azacycle
- Monocarboxylic acid or derivatives
- Hydrocarbon derivative
- Organic oxygen compound
- Organic oxide
- Organic nitrogen compound
- Organooxygen compound
- Organopnictogen compound
- Organonitrogen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0kai-5009000000-6681d3289255ce397469 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (3 TMS) - 70eV, Positive | splash10-004i-9510760000-2358e8c404b7a8c37784 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-01q9-0019000000-3ca16854d8e49244f3e5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-01qi-0459000000-d2a5e5f146a00010dcc1 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0019-3973000000-62d43753172e8b7cfeeb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0002-0019000000-4eeccb310308d12ab431 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0ug1-0059000000-ffd7a1003ebd6533ea18 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-2392000000-afc541c6296f4be502a9 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0030215 |
|---|
| FooDB ID | FDB002034 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00029986 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 28285416 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 24898872 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|