| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 00:47:19 UTC |
|---|
| Update Date | 2016-11-09 01:18:47 UTC |
|---|
| Accession Number | CHEM028909 |
|---|
| Identification |
|---|
| Common Name | Apo-12'-violaxanthal |
|---|
| Class | Small Molecule |
|---|
| Description | Apo-12'-violaxanthal is found in fruits. Apo-12'-violaxanthal is isolated from plums Prunus domestic |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
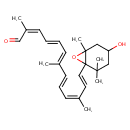
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 5,6-Epoxy-5,6-dihydro-3b-hydroxy-12'-apo-b-caroten-12'-al | HMDB |
|
|---|
| Chemical Formula | C25H34O3 |
|---|
| Average Molecular Mass | 382.536 g/mol |
|---|
| Monoisotopic Mass | 382.251 g/mol |
|---|
| CAS Registry Number | Not Available |
|---|
| IUPAC Name | (2Z,4E,6E,8E,10Z,12E)-13-{4-hydroxy-2,2,6-trimethyl-7-oxabicyclo[4.1.0]heptan-1-yl}-2,7,11-trimethyltrideca-2,4,6,8,10,12-hexaenal |
|---|
| Traditional Name | (2Z,4E,6E,8E,10Z,12E)-13-{4-hydroxy-2,2,6-trimethyl-7-oxabicyclo[4.1.0]heptan-1-yl}-2,7,11-trimethyltrideca-2,4,6,8,10,12-hexaenal |
|---|
| SMILES | C\C(C=O)=C\C=C\C=C(/C)\C=C\C=C(\C)/C=C/C12OC1(C)CC(O)CC2(C)C |
|---|
| InChI Identifier | InChI=1S/C25H34O3/c1-19(10-7-8-11-21(3)18-26)12-9-13-20(2)14-15-25-23(4,5)16-22(27)17-24(25,6)28-25/h7-15,18,22,27H,16-17H2,1-6H3/b8-7+,12-9+,15-14+,19-10+,20-13-,21-11- |
|---|
| InChI Key | CAXVJDRXJFKYQP-RXCALXPUSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as diterpenoids. These are terpene compounds formed by four isoprene units. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Prenol lipids |
|---|
| Sub Class | Diterpenoids |
|---|
| Direct Parent | Diterpenoids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Diterpenoid
- Oxepane
- Alpha,beta-unsaturated aldehyde
- Cyclic alcohol
- Enal
- Secondary alcohol
- Dialkyl ether
- Oxacycle
- Oxirane
- Ether
- Organoheterocyclic compound
- Hydrocarbon derivative
- Alcohol
- Organic oxide
- Aldehyde
- Organic oxygen compound
- Organooxygen compound
- Carbonyl group
- Aliphatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aliphatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-014l-5019000000-859646253189c493ea3f | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (1 TMS) - 70eV, Positive | splash10-000i-9723800000-ae5c58f3bf540052cb74 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0159-1239000000-e8b8b333ae0c5b0acd97 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0292-1975000000-0f548ce3175265edf642 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0aor-9710000000-554899cbb25bbbb2f6da | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-001i-0009000000-1319620aeda5472d1bf6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-01q9-0009000000-6904a5981f74e0925792 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0pca-8965000000-301294cb2f70e9d24029 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-016r-0129000000-2cf095b56f4d41d41ebe | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0159-1769000000-bdc8fec26ecee587b947 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-014i-4901000000-de9a78be90f77437144f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0f89-0209000000-f0a239a1dd8ae4de7789 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0udi-0109000000-984c640e5b99df56359c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0k9b-0649000000-4842e9b6e5868a43393d | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0034953 |
|---|
| FooDB ID | FDB013546 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 35013802 |
|---|
| ChEBI ID | 171885 |
|---|
| PubChem Compound ID | 131751642 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|