| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 00:46:14 UTC |
|---|
| Update Date | 2016-11-09 01:18:46 UTC |
|---|
| Accession Number | CHEM028879 |
|---|
| Identification |
|---|
| Common Name | Lettowianthine |
|---|
| Class | Small Molecule |
|---|
| Description | Alkaloid from Annona glabra (pond apple). Lettowianthine is found in alcoholic beverages and fruits. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
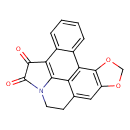
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Annobraine | HMDB | | Lettowianthine | MeSH |
|
|---|
| Chemical Formula | C19H11NO4 |
|---|
| Average Molecular Mass | 317.295 g/mol |
|---|
| Monoisotopic Mass | 317.069 g/mol |
|---|
| CAS Registry Number | Not Available |
|---|
| IUPAC Name | 3,5-dioxa-11-azahexacyclo[9.9.2.0²,⁶.0⁸,²¹.0¹⁴,²².0¹⁵,²⁰]docosa-1(21),2(6),7,14(22),15,17,19-heptaene-12,13-dione |
|---|
| Traditional Name | 3,5-dioxa-11-azahexacyclo[9.9.2.0²,⁶.0⁸,²¹.0¹⁴,²².0¹⁵,²⁰]docosa-1(21),2(6),7,14(22),15,17,19-heptaene-12,13-dione |
|---|
| SMILES | O=C1N2CCC3=CC4=C(OCO4)C4=C3C2=C(C1=O)C1=CC=CC=C41 |
|---|
| InChI Identifier | InChI=1S/C19H11NO4/c21-17-15-11-4-2-1-3-10(11)14-13-9(7-12-18(14)24-8-23-12)5-6-20(16(13)15)19(17)22/h1-4,7H,5-6,8H2 |
|---|
| InChI Key | FMLHJJVSHOCVAU-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as aporphines. These are quinoline alkaloids containing the dibenzo[de,g]quinoline ring system or a dehydrogenated derivative thereof. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Alkaloids and derivatives |
|---|
| Class | Aporphines |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Aporphines |
|---|
| Alternative Parents | |
|---|
| Substituents | - Aporphine
- Benzoquinoline
- Phenanthrene
- Naphthalene
- Quinoline
- Benzodioxole
- Indole or derivatives
- Aryl ketone
- Benzenoid
- Vinylogous amide
- Tertiary carboxylic acid amide
- Carboxamide group
- Ketone
- Lactam
- Acetal
- Carboxylic acid derivative
- Oxacycle
- Azacycle
- Organoheterocyclic compound
- Hydrocarbon derivative
- Organic nitrogen compound
- Organic oxide
- Carbonyl group
- Organic oxygen compound
- Organopnictogen compound
- Organonitrogen compound
- Organooxygen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-000i-0092000000-9e89e9fe7d52ccf28ba9 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014i-0019000000-95327c7827d0194b8995 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-014i-0029000000-6d71d316f416721f4ece | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0310-0090000000-3c98d1efe4b186c050af | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-014i-0009000000-8d463ca6bf3eeb8115dd | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-02t9-0096000000-a84d45628a8f2ec4689e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-08g0-0090000000-88db4609af2a4d78ab7c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-014i-0009000000-cdb697e9f2878552c94c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-014i-0009000000-0c180e871813a2016735 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0wmr-0098000000-ef5fa2a1d42687d1bc6e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-014i-0009000000-7a22534e87f602b14ab3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-014i-0009000000-7a22534e87f602b14ab3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-02tc-0094000000-63db42333d87563cf792 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0034923 |
|---|
| FooDB ID | FDB013509 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00027280 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 10216687 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 21593830 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|