| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 00:45:27 UTC |
|---|
| Update Date | 2016-11-09 01:18:46 UTC |
|---|
| Accession Number | CHEM028861 |
|---|
| Identification |
|---|
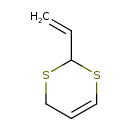
| Common Name | 2-Vinyl-4H-1,3-dithiine |
|---|
| Class | Small Molecule |
|---|
| Description | 2-Vinyl-4H-1,3-dithiine is found in onion-family vegetables. 2-Vinyl-4H-1,3-dithiine is present in garlic (Allium sativum) and caucas (Allium victorialis) but not in onion or rakky |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 1-Ethyltridecyl 3-bromobenzoate | HMDB | | 2-Ethenyl-4H-1,3-dithiin | HMDB | | 2-Ethenyl-4H-1,3-dithiine, 9ci | HMDB | | 2-Vdtii | HMDB | | 2-Vinyl-(4H)-1,3-dithiine | HMDB | | 2-Vinyl-4(H)-1,3-dithiin | HMDB | | 2-Vinyl-4H-1,3-dithiin | HMDB | | 2-Vinyl-4H-1,3-dithin | HMDB | | 2-Vinyl-[4H]-1,3-dithin | HMDB | | 3-Bromobenzoic acid, 3-pentadecyl ester | HMDB | | 4H-1,3-Dithiin, 2-ethenyl | HMDB |
|
|---|
| Chemical Formula | C6H8S2 |
|---|
| Average Molecular Mass | 144.258 g/mol |
|---|
| Monoisotopic Mass | 144.007 g/mol |
|---|
| CAS Registry Number | 80028-57-5 |
|---|
| IUPAC Name | 2-ethenyl-2,4-dihydro-1,3-dithiine |
|---|
| Traditional Name | 2-ethenyl-2,4-dihydro-1,3-dithiine |
|---|
| SMILES | C=CC1SCC=CS1 |
|---|
| InChI Identifier | InChI=1S/C6H8S2/c1-2-6-7-4-3-5-8-6/h2-4,6H,1,5H2 |
|---|
| InChI Key | XUKBDTUPIIADOP-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as dithiins. Dithiins are compounds comprising a dithiin ring, which is an unsaturated six-member heterocycle containing four carbon atoms, two sulfur atoms and two double bonds. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organoheterocyclic compounds |
|---|
| Class | Dithiins |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Dithiins |
|---|
| Alternative Parents | |
|---|
| Substituents | - 1,3-dithiin
- Thioacetal
- Thioenolether
- Dialkylthioether
- Thioether
- Hydrocarbon derivative
- Aliphatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aliphatic heteromonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0076-9300000000-427ec03e23132d179c77 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0002-0900000000-3e4031f35064cbb84c6c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0002-9400000000-3f55c8c9bcb5d7f640b8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-00di-9200000000-b8c078f742906e31bf23 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0k97-7900000000-d5450a6d81360c9060b7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00di-9000000000-4f1ad5366369dc5b28f9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-00di-9000000000-63208c7fa296fbcdcd5a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-006x-5900000000-aee8163659956229a887 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00dl-9400000000-990c5bd04132b963aca6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-00dl-9800000000-4bcdcb7bb21fc0af6075 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0002-3900000000-e3821642d6b887647ed2 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-007a-9200000000-27e6f3875eaaa018745f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-05g0-9000000000-f8425ec58d9ee201de03 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0034901 |
|---|
| FooDB ID | FDB013480 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00058205 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | CPD-9295 |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 117630 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 133337 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|