| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 00:32:14 UTC |
|---|
| Update Date | 2016-11-09 01:18:43 UTC |
|---|
| Accession Number | CHEM028557 |
|---|
| Identification |
|---|
| Common Name | 16-Acetylpriverogenin A |
|---|
| Class | Small Molecule |
|---|
| Description | 16-Acetylpriverogenin A is found in herbs and spices. Sapogenin from Primula veris (cowslip |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
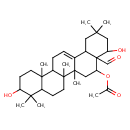
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 4a-Formyl-4,10-dihydroxy-2,2,6a,6b,9,9,12a-heptamethyl-1,2,3,4,4a,5,6,6a,6b,7,8,8a,9,10,11,12,12a,12b,13,14b-icosahydropicen-5-yl acetic acid | HMDB |
|
|---|
| Chemical Formula | C32H50O5 |
|---|
| Average Molecular Mass | 514.736 g/mol |
|---|
| Monoisotopic Mass | 514.366 g/mol |
|---|
| CAS Registry Number | 26339-85-5 |
|---|
| IUPAC Name | 4a-formyl-4,10-dihydroxy-2,2,6a,6b,9,9,12a-heptamethyl-1,2,3,4,4a,5,6,6a,6b,7,8,8a,9,10,11,12,12a,12b,13,14b-icosahydropicen-5-yl acetate |
|---|
| Traditional Name | 4a-formyl-4,10-dihydroxy-2,2,6a,6b,9,9,12a-heptamethyl-1,3,4,5,6,7,8,8a,10,11,12,12b,13,14b-tetradecahydropicen-5-yl acetate |
|---|
| SMILES | CC(=O)OC1CC2(C)C(=CCC3C4(C)CCC(O)C(C)(C)C4CCC23C)C2CC(C)(C)CC(O)C12C=O |
|---|
| InChI Identifier | InChI=1S/C32H50O5/c1-19(34)37-26-17-31(8)20(21-15-27(2,3)16-25(36)32(21,26)18-33)9-10-23-29(6)13-12-24(35)28(4,5)22(29)11-14-30(23,31)7/h9,18,21-26,35-36H,10-17H2,1-8H3 |
|---|
| InChI Key | RVLOLIBRTUMCPC-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as triterpenoids. These are terpene molecules containing six isoprene units. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Prenol lipids |
|---|
| Sub Class | Triterpenoids |
|---|
| Direct Parent | Triterpenoids |
|---|
| Alternative Parents | |
|---|
| Substituents | - Triterpenoid
- Cyclic alcohol
- Secondary alcohol
- Carboxylic acid ester
- Monocarboxylic acid or derivatives
- Carboxylic acid derivative
- Organic oxygen compound
- Organic oxide
- Hydrocarbon derivative
- Organooxygen compound
- Carbonyl group
- Aldehyde
- Alcohol
- Aliphatic homopolycyclic compound
|
|---|
| Molecular Framework | Aliphatic homopolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0005-2010900000-f3345b5d05c912e3dd11 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (2 TMS) - 70eV, Positive | splash10-0006-3011129000-2f8a9fd1863eccd72f24 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00mk-0000910000-6ba4c58e6994c781fea6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0a71-0000900000-57891dfa1905560d5732 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0abi-2051900000-ca01cef02b5f0421af18 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03di-1000960000-c443eb9cd7b424ac3e9d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-11ba-2000920000-5b4a38a81c8587eb8b95 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4l-5000900000-4211b6d054bc51c0f1a4 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-08fr-6000290000-69c4999a2e0852e691b3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0bt9-9000650000-858b292c39cba9b86159 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0kal-6900400000-c8b908840a9b6afa00af | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0170-0000950000-389e0ef84fda66b18799 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-05r0-0709810000-62923e4a6cba0f6d56ae | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-00p4-2943300000-2843d6c8a3cf343651b8 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0034529 |
|---|
| FooDB ID | FDB013030 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 532468 |
|---|
| ChEBI ID | 175898 |
|---|
| PubChem Compound ID | 612545 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|