| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 00:30:33 UTC |
|---|
| Update Date | 2016-11-09 01:18:42 UTC |
|---|
| Accession Number | CHEM028507 |
|---|
| Identification |
|---|
| Common Name | Curvacin A |
|---|
| Class | Small Molecule |
|---|
| Description | Not Available |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
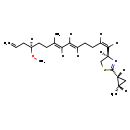
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| Curacin C | MeSH | | Curacin b | MeSH |
|
|---|
| Chemical Formula | C23H35NOS |
|---|
| Average Molecular Mass | 373.600 g/mol |
|---|
| Monoisotopic Mass | 373.244 g/mol |
|---|
| CAS Registry Number | 146239-44-3 |
|---|
| IUPAC Name | (4R)-4-[(1Z,5E,7E,11R)-11-methoxy-8-methyltetradeca-1,5,7,13-tetraen-1-yl]-2-[(1R,2S)-2-methylcyclopropyl]-4,5-dihydro-1,3-thiazole |
|---|
| Traditional Name | (4R)-4-[(1Z,5E,7E,11R)-11-methoxy-8-methyltetradeca-1,5,7,13-tetraen-1-yl]-2-[(1R,2S)-2-methylcyclopropyl]-4,5-dihydro-1,3-thiazole |
|---|
| SMILES | [H]\C(CC\C([H])=C(\[H])[C@]1([H])CSC(=N1)[C@]1([H])C[C@]1([H])C)=C(\[H])/C(/[H])=C(\C)CC[C@]([H])(CC=C)OC |
|---|
| InChI Identifier | InChI=1S/C23H35NOS/c1-5-11-21(25-4)15-14-18(2)12-9-7-6-8-10-13-20-17-26-23(24-20)22-16-19(22)3/h5,7,9-10,12-13,19-22H,1,6,8,11,14-17H2,2-4H3/b9-7+,13-10-,18-12+/t19-,20+,21-,22+/m0/s1 |
|---|
| InChI Key | LUEYTMPPCOCKBX-KWYHTCOPSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as imidothiolactones. These are analogs of imidolactones where the oxygen atom is replaced by a sulfur atom. They have the general structure RC(=N)SR' where the central carbon atom is part of a ring. R,R' = organyl group. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Organosulfur compounds |
|---|
| Class | Imidothioesters |
|---|
| Sub Class | Imidothiolactones |
|---|
| Direct Parent | Imidothiolactones |
|---|
| Alternative Parents | |
|---|
| Substituents | - Imidothiolactone
- Meta-thiazoline
- Azacycle
- Organoheterocyclic compound
- Organic 1,3-dipolar compound
- Propargyl-type 1,3-dipolar organic compound
- Ether
- Dialkyl ether
- Organic nitrogen compound
- Organic oxygen compound
- Organopnictogen compound
- Hydrocarbon derivative
- Organooxygen compound
- Organonitrogen compound
- Aliphatic heteromonocyclic compound
|
|---|
| Molecular Framework | Aliphatic heteromonocyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-006t-4249000000-f61f36a7de5dea112725 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0089-2779000000-a2191905078bfee421f8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-00ke-9620000000-d30b24ae2d1171a7267e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0229-7619000000-8a69a27fa087dfac9598 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-001i-4009000000-0450e7889df8b552063e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a5a-9000000000-d186c4cfdd907f58f9ee | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0r03-2095000000-f1b9db835985613f5199 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0kmi-3193000000-fba55bebb8936010af54 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0159-8930000000-f8e0172925b0a8d3b4c7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-00di-0019000000-81485029a44ee195b69c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-00di-3139000000-d8f6945e7ec887726f0e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-001r-4091000000-3fa9d2b624323e221c6f | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0303393 |
|---|
| FooDB ID | FDB012935 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00027627 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 4445239 |
|---|
| ChEBI ID | 3961 |
|---|
| PubChem Compound ID | 5281967 |
|---|
| Kegg Compound ID | C11689 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | Not Available |
|---|