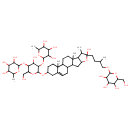| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 00:16:11 UTC |
|---|
| Update Date | 2016-11-09 01:18:37 UTC |
|---|
| Accession Number | CHEM028174 |
|---|
| Identification |
|---|
| Common Name | Protodioscin |
|---|
| Class | Small Molecule |
|---|
| Description | From Asparagus officinalis (asparagus)
Protodioscin is a steroidal saponin compound found in a number of plant species, most notably in the Tribulus, Trigonella and Dioscorea families. It is best known as the putative active component of the herbal aphrodisiac plant Tribulus terrestris. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| NSC 698796 | HMDB | | Saponin c? | HMDB | | Protoneodioscin | HMDB | | Protodioscin | MeSH |
|
|---|
| Chemical Formula | C51H84O22 |
|---|
| Average Molecular Mass | 1049.200 g/mol |
|---|
| Monoisotopic Mass | 1048.545 g/mol |
|---|
| CAS Registry Number | 55056-80-9 |
|---|
| IUPAC Name | 2-[(4-hydroxy-6-{[6-hydroxy-7,9,13-trimethyl-6-(3-methyl-4-{[3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}butyl)-5-oxapentacyclo[10.8.0.0²,⁹.0⁴,⁸.0¹³,¹⁸]icos-18-en-16-yl]oxy}-2-(hydroxymethyl)-5-[(3,4,5-trihydroxy-6-methyloxan-2-yl)oxy]oxan-3-yl)oxy]-6-methyloxane-3,4,5-triol |
|---|
| Traditional Name | 2-[(4-hydroxy-6-{[6-hydroxy-7,9,13-trimethyl-6-(3-methyl-4-{[3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}butyl)-5-oxapentacyclo[10.8.0.0²,⁹.0⁴,⁸.0¹³,¹⁸]icos-18-en-16-yl]oxy}-2-(hydroxymethyl)-5-[(3,4,5-trihydroxy-6-methyloxan-2-yl)oxy]oxan-3-yl)oxy]-6-methyloxane-3,4,5-triol |
|---|
| SMILES | CC(CCC1(O)OC2CC3C4CC=C5CC(CCC5(C)C4CCC3(C)C2C1C)OC1OC(CO)C(OC2OC(C)C(O)C(O)C2O)C(O)C1OC1OC(C)C(O)C(O)C1O)COC1OC(CO)C(O)C(O)C1O |
|---|
| InChI Identifier | InChI=1S/C51H84O22/c1-20(19-65-45-39(60)38(59)35(56)30(17-52)69-45)9-14-51(64)21(2)32-29(73-51)16-28-26-8-7-24-15-25(10-12-49(24,5)27(26)11-13-50(28,32)6)68-48-44(72-47-41(62)37(58)34(55)23(4)67-47)42(63)43(31(18-53)70-48)71-46-40(61)36(57)33(54)22(3)66-46/h7,20-23,25-48,52-64H,8-19H2,1-6H3 |
|---|
| InChI Key | LVTJOONKWUXEFR-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as steroidal saponins. These are saponins in which the aglycone moiety is a steroid. The steroidal aglycone is usually a spirostane, furostane, spirosolane, solanidane, or curcubitacin derivative. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Lipids and lipid-like molecules |
|---|
| Class | Steroids and steroid derivatives |
|---|
| Sub Class | Steroidal glycosides |
|---|
| Direct Parent | Steroidal saponins |
|---|
| Alternative Parents | Not Available |
|---|
| Substituents | Not Available |
|---|
| Molecular Framework | Aliphatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-001a-6110180693-bed8bdf4b94c97fe4b5f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-052s-1220370951-5c7ca73832f61158be95 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a4r-3213250930-ac533d5102bcd8103db8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0h01-9400030353-a9291f2865c3ddfa4fcf | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-03fu-5900051132-5a00f7647168ce7080a6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-03dl-4900150301-1464cb3c4903a6198a91 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-002k-9700000040-2bb0f9418b0b7bfeb118 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0kbb-6903020823-766ae72a06877000fa44 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0a6s-9610010122-ce3b33eb0761e26155d8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0002-9200000012-a291c3f18415dda5b1c5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-052e-9400000013-60abf0511314048ebd01 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0k96-9200000622-835e40aeed25a97166c8 | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum | | 1D NMR | 13C NMR Spectrum | Not Available | Spectrum | | 1D NMR | 1H NMR Spectrum | Not Available | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0034062 |
|---|
| FooDB ID | FDB012311 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00003596 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Protodioscin |
|---|
| Chemspider ID | 437743 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 500375 |
|---|
| Kegg Compound ID | C08907 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|