| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 00:15:40 UTC |
|---|
| Update Date | 2016-11-09 01:18:37 UTC |
|---|
| Accession Number | CHEM028161 |
|---|
| Identification |
|---|
| Common Name | Psoralidin |
|---|
| Class | Small Molecule |
|---|
| Description | A member of the class of coumestans that is coumestan substituted by hydroxy groups at positions 3 and 9 and a prenyl group at position 2 respectively. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
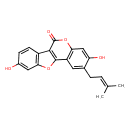
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 3,9-Dihydroxy-2-prenylcoumestan | ChEBI |
|
|---|
| Chemical Formula | C20H16O5 |
|---|
| Average Molecular Mass | 336.338 g/mol |
|---|
| Monoisotopic Mass | 336.100 g/mol |
|---|
| CAS Registry Number | 18642-23-4 |
|---|
| IUPAC Name | 5,14-dihydroxy-4-(3-methylbut-2-en-1-yl)-8,17-dioxatetracyclo[8.7.0.0²,⁷.0¹¹,¹⁶]heptadeca-1(10),2(7),3,5,11(16),12,14-heptaen-9-one |
|---|
| Traditional Name | psoralidin |
|---|
| SMILES | CC(C)=CCC1=CC2=C(OC(=O)C3=C2OC2=C3C=CC(O)=C2)C=C1O |
|---|
| InChI Identifier | InChI=1S/C20H16O5/c1-10(2)3-4-11-7-14-17(9-15(11)22)25-20(23)18-13-6-5-12(21)8-16(13)24-19(14)18/h3,5-9,21-22H,4H2,1-2H3 |
|---|
| InChI Key | YABIJLLNNFURIJ-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as coumestans. These are polycyclic aromatic compounds containing a coumestan moiety, which consists of a benzoxole fused to a chromen-2-one to form 1-Benzoxolo[3,2-c]chromen-6-one. They are oxidation products of pterocarpan. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Phenylpropanoids and polyketides |
|---|
| Class | Isoflavonoids |
|---|
| Sub Class | Coumestans |
|---|
| Direct Parent | Coumestans |
|---|
| Alternative Parents | |
|---|
| Substituents | - Coumestan
- Angular furanocoumarin
- Furanocoumarin
- Coumarin
- Benzopyran
- 1-benzopyran
- Benzofuran
- Furopyran
- 1-hydroxy-2-unsubstituted benzenoid
- Pyranone
- Pyran
- Benzenoid
- Furan
- Heteroaromatic compound
- Lactone
- Organoheterocyclic compound
- Oxacycle
- Organooxygen compound
- Hydrocarbon derivative
- Organic oxide
- Organic oxygen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0a6u-3649000000-fb4bed2219264a94b2c0 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (2 TMS) - 70eV, Positive | splash10-06di-2021900000-af5a7fcfd2abc930bab8 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-000i-0029000000-611c9c21982e8c8417bc | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00m0-5097000000-522ed689eb20e83a6060 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0159-9160000000-7eabc2db1056ba50d5b1 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-000i-0029000000-7390a95ac6006bbd4672 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-000i-0039000000-6317b2200c19c15fe546 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a5c-2892000000-2dc8e8c23fe68b07d783 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-000i-0009000000-dc81427ba733f47873d1 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-000i-0019000000-acb098f91f6b555322c7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0umr-0190000000-a3b3ed9e9206503a2435 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-000i-0019000000-af3d0665d7147a8a9b18 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-001r-0095000000-7b1aff3dc2da88d695d5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0far-0090000000-319a7d07214529aec4af | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0034050 |
|---|
| FooDB ID | FDB012294 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00002566 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Psoralidin |
|---|
| Chemspider ID | 4445118 |
|---|
| ChEBI ID | 8616 |
|---|
| PubChem Compound ID | 5281806 |
|---|
| Kegg Compound ID | C10523 |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|