| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 00:14:17 UTC |
|---|
| Update Date | 2016-11-09 01:18:37 UTC |
|---|
| Accession Number | CHEM028126 |
|---|
| Identification |
|---|
| Common Name | 6-Hydroxy-9,9-dimethyl-5-(3-methyl-1-oxobutyl)-1-propyl-3H,9H-[1,2]-dioxolo[3',4':4,5]furo[2,3-f][1]benzopyran-3-one |
|---|
| Class | Small Molecule |
|---|
| Description | 6-Hydroxy-9,9-dimethyl-5-(3-methyl-1-oxobutyl)-1-propyl-3H,9H-[1,2]-dioxolo[3',4':4,5]furo[2,3-f][1]benzopyran-3-one is found in fruits. 6-Hydroxy-9,9-dimethyl-5-(3-methyl-1-oxobutyl)-1-propyl-3H,9H-[1,2]-dioxolo[3',4':4,5]furo[2,3-f][1]benzopyran-3-one is isolated from Mammea americana (mamey). 6-Hydroxy-9,9-dimethyl-5-(3-methyl-1-oxobutyl)-1-propyl-3H,9H-[1,2]-dioxolo[3',4':4,5]furo[2,3-f][1]benzopyran-3-one is an oxidation produced of Mammein. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
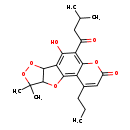
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 1-Pyridin-2-yl-9H-beta-carboline | HMDB |
|
|---|
| Chemical Formula | C22H26O7 |
|---|
| Average Molecular Mass | 402.438 g/mol |
|---|
| Monoisotopic Mass | 402.168 g/mol |
|---|
| CAS Registry Number | 30390-08-0 |
|---|
| IUPAC Name | 9-hydroxy-14,14-dimethyl-8-(3-methylbutanoyl)-3-propyl-6,12,13,16-tetraoxatetracyclo[8.6.0.0²,⁷.0¹¹,¹⁵]hexadeca-1,3,7,9-tetraen-5-one |
|---|
| Traditional Name | 9-hydroxy-14,14-dimethyl-8-(3-methylbutanoyl)-3-propyl-6,12,13,16-tetraoxatetracyclo[8.6.0.0²,⁷.0¹¹,¹⁵]hexadeca-1,3,7,9-tetraen-5-one |
|---|
| SMILES | CCCC1=CC(=O)OC2=C(C(=O)CC(C)C)C(O)=C3C4OOC(C)(C)C4OC3=C12 |
|---|
| InChI Identifier | InChI=1S/C22H26O7/c1-6-7-11-9-13(24)26-18-14(11)19-16(17(25)15(18)12(23)8-10(2)3)20-21(27-19)22(4,5)29-28-20/h9-10,20-21,25H,6-8H2,1-5H3 |
|---|
| InChI Key | HXNFGNDKYRUBRV-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as angular furanocoumarins. These are furanocoumarins, with a structure characterized by a furan ring angularly fused to a coumarin. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Phenylpropanoids and polyketides |
|---|
| Class | Coumarins and derivatives |
|---|
| Sub Class | Furanocoumarins |
|---|
| Direct Parent | Angular furanocoumarins |
|---|
| Alternative Parents | |
|---|
| Substituents | - Angular furanocoumarin
- Butyrophenone
- Benzopyran
- 1-benzopyran
- Coumaran
- Aryl alkyl ketone
- Aryl ketone
- Alkyl aryl ether
- Pyranone
- Benzenoid
- Pyran
- Ortho-dioxolane
- Heteroaromatic compound
- Vinylogous acid
- Dialkyl peroxide
- Ketone
- Lactone
- Ether
- Oxacycle
- Organoheterocyclic compound
- Organic oxide
- Organic oxygen compound
- Hydrocarbon derivative
- Organooxygen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0a4i-6119000000-5172fb428761cfb524db | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (1 TMS) - 70eV, Positive | splash10-0a4i-8110900000-0311381745bc07922e99 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0udi-2009700000-6331d856b247da61d117 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-054k-4029100000-464ff2ae0227237686b0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-054o-8095000000-ac11573ff3ba41b55f3e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-3005900000-08c52dbc8d195066d697 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0pvi-7029300000-094e88781c0a2b559302 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0a4i-9041000000-048328fd13f5b38dcd73 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0udi-0000900000-fa1aba5cde120fbcce87 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0udi-0009800000-5e2b9315550679a12ff8 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-014i-1019000000-66d63f3753cb5d21cd17 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-0udi-0000900000-694f72938bd541622142 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0udi-0006900000-c5e16c5c60c57914f630 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0ktf-4109000000-16707c561ae047a79fe4 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0034016 |
|---|
| FooDB ID | FDB012254 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 35013686 |
|---|
| ChEBI ID | 175987 |
|---|
| PubChem Compound ID | 131751512 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|