| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 00:10:04 UTC |
|---|
| Update Date | 2016-11-09 01:18:35 UTC |
|---|
| Accession Number | CHEM028024 |
|---|
| Identification |
|---|
| Common Name | 4,5-Dimethoxy-1,2-benzenedicarboxylic acid |
|---|
| Class | Small Molecule |
|---|
| Description | 4,5-Dimethoxy-1,2-benzenedicarboxylic acid is a degradation product produced of many alkaloids. It is isolated from poppy straw (Papaver somniferum). |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
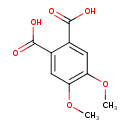
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 4,5-Dimethoxy-1,2-benzenedicarboxylate | Generator | | 4, 5-Dimethoxy-1,2-benzenedicarboxylic acid | HMDB | | 4,5-Dimethoxy-phthalic acid | HMDB | | 4,5-Dimethoxyphthalic acid | HMDB | | m-Hemipic acid | HMDB | | m-Hemipinic acid | HMDB | | Metahemipic acid | HMDB | | Veratrole-4,5-dicarboxylic acid | HMDB | | 4,5-Dimethoxybenzene-1,2-dicarboxylate | Generator |
|
|---|
| Chemical Formula | C10H10O6 |
|---|
| Average Molecular Mass | 226.183 g/mol |
|---|
| Monoisotopic Mass | 226.048 g/mol |
|---|
| CAS Registry Number | 577-68-4 |
|---|
| IUPAC Name | 4,5-dimethoxybenzene-1,2-dicarboxylic acid |
|---|
| Traditional Name | 4,5-dimethoxybenzene-1,2-dicarboxylic acid |
|---|
| SMILES | COC1=C(OC)C=C(C(O)=O)C(=C1)C(O)=O |
|---|
| InChI Identifier | InChI=1S/C10H10O6/c1-15-7-3-5(9(11)12)6(10(13)14)4-8(7)16-2/h3-4H,1-2H3,(H,11,12)(H,13,14) |
|---|
| InChI Key | SKBDLRWFSRLIPP-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as p-methoxybenzoic acids and derivatives. These are benzoic acids in which the hydrogen atom at position 4 of the benzene ring is replaced by a methoxy group. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Benzenoids |
|---|
| Class | Benzene and substituted derivatives |
|---|
| Sub Class | Benzoic acids and derivatives |
|---|
| Direct Parent | P-methoxybenzoic acids and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - P-methoxybenzoic acid or derivatives
- M-methoxybenzoic acid or derivatives
- O-dimethoxybenzene
- Dimethoxybenzene
- Benzoic acid
- Phenoxy compound
- Anisole
- Methoxybenzene
- Phenol ether
- Benzoyl
- Alkyl aryl ether
- Dicarboxylic acid or derivatives
- Carboxylic acid derivative
- Carboxylic acid
- Ether
- Organooxygen compound
- Hydrocarbon derivative
- Organic oxygen compound
- Organic oxide
- Aromatic homomonocyclic compound
|
|---|
| Molecular Framework | Aromatic homomonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0560-2970000000-1b8bed387dae5bb189de | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (2 TMS) - 70eV, Positive | splash10-00di-9054000000-784bf01cd33a0dab1216 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-004i-0090000000-0e2f409a57fc2c2b2ae2 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-004i-0290000000-8dc5242bc07e1f15a094 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0059-0910000000-30117e1cf8d87a81e3c2 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-0390000000-dfc19af6ee36612dc0fb | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-0059-0950000000-266875143116075421a9 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0kl9-1900000000-6cd880fa0753698c961f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0a6r-0090000000-8de715c1593d4b288724 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-056r-0390000000-a9c4942ece7399dc166a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-014j-9710000000-6f8aee5850ec7ed6269a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-001i-0910000000-bb4488dc60ef1f78e397 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-000i-0910000000-a9cb9ed3672903b204f5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-00kr-3900000000-f813e190d865c6b2470d | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0033893 |
|---|
| FooDB ID | FDB012088 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00058139 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 256696 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 290988 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|