| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-26 00:07:22 UTC |
|---|
| Update Date | 2016-11-09 01:18:34 UTC |
|---|
| Accession Number | CHEM027964 |
|---|
| Identification |
|---|
| Common Name | 4-Hydroxy-2,3,9-trimethoxypterocarpan |
|---|
| Class | Small Molecule |
|---|
| Description | 4-Hydroxy-2,3,9-trimethoxypterocarpan is found in common pea. 4-Hydroxy-2,3,9-trimethoxypterocarpan is isolated from Pisum sativum (pea). |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
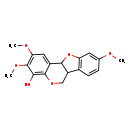
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 6a,11a-dihydro-2,3,9-Trimethoxy-6H-benzofuro[3,2-c][1]benzopyran-4-ol, 9ci | HMDB |
|
|---|
| Chemical Formula | C18H18O6 |
|---|
| Average Molecular Mass | 330.332 g/mol |
|---|
| Monoisotopic Mass | 330.110 g/mol |
|---|
| CAS Registry Number | 56841-82-8 |
|---|
| IUPAC Name | 4,5,14-trimethoxy-8,17-dioxatetracyclo[8.7.0.0²,⁷.0¹¹,¹⁶]heptadeca-2,4,6,11(16),12,14-hexaen-6-ol |
|---|
| Traditional Name | 4,5,14-trimethoxy-8,17-dioxatetracyclo[8.7.0.0²,⁷.0¹¹,¹⁶]heptadeca-2,4,6,11(16),12,14-hexaen-6-ol |
|---|
| SMILES | COC1=CC2=C(C=C1)C1COC3=C(O)C(OC)=C(OC)C=C3C1O2 |
|---|
| InChI Identifier | InChI=1S/C18H18O6/c1-20-9-4-5-10-12-8-23-17-11(16(12)24-13(10)6-9)7-14(21-2)18(22-3)15(17)19/h4-7,12,16,19H,8H2,1-3H3 |
|---|
| InChI Key | APDOGYRSSCEXNB-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as pterocarpans. These are benzo-pyrano-furano-benzene compounds, containing the 6H-[1]benzofuro[3,2-c]chromene skeleton. They are derivatives of isoflavonoids. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Phenylpropanoids and polyketides |
|---|
| Class | Isoflavonoids |
|---|
| Sub Class | Furanoisoflavonoids |
|---|
| Direct Parent | Pterocarpans |
|---|
| Alternative Parents | |
|---|
| Substituents | - Pterocarpan
- Isoflavanol
- Isoflavan
- Chromane
- 1-benzopyran
- Benzopyran
- Coumaran
- Benzofuran
- Anisole
- 1-hydroxy-4-unsubstituted benzenoid
- Alkyl aryl ether
- Benzenoid
- Oxacycle
- Organoheterocyclic compound
- Ether
- Organic oxygen compound
- Hydrocarbon derivative
- Organooxygen compound
- Aromatic heteropolycyclic compound
|
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-0gba-0739000000-1612946b221d02cc0048 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (1 TMS) - 70eV, Positive | splash10-0079-1129000000-50a1ad62992542def8ab | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-00lr-0509000000-1fb33ec9c409a691f00a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-00lr-0629000000-0ff70174fb6e16fab96c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-03y1-2910000000-f098e6385ff8e7a72570 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-0009000000-38d5cd86001d90f602bf | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-01ta-0069000000-9f5a67b20b156424917c | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-06wc-2390000000-dfbed0c21ebeb62a9062 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-001i-0009000000-2160799bc7c624fd0646 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-001i-0009000000-b00061307acdcb1e67c0 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-03xr-0693000000-208fd85a933bc5628f9d | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-004i-0009000000-de11969ae6615b28f3d7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-016s-0094000000-4d8d06564b41844767d6 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-05mx-2191000000-b9eda0f9598c4758bd08 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0033817 |
|---|
| FooDB ID | FDB011981 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00009633 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 24843022 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 23036629 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|