| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 23:54:05 UTC |
|---|
| Update Date | 2016-11-09 01:18:30 UTC |
|---|
| Accession Number | CHEM027675 |
|---|
| Identification |
|---|
| Common Name | Caffeoylferuloylspermidine |
|---|
| Class | Small Molecule |
|---|
| Description | Caffeoylferuloylspermidine is found in nuts such as common hazelnut. Caffeoylferuloylspermidine is an alkaloid from the pollen of Corylus avellana (filbert). |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
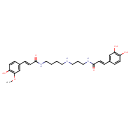
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| (2E)-3-(3,4-Dihydroxyphenyl)-N-{3-[(4-{[(2E)-1-hydroxy-3-(4-hydroxy-3-methoxyphenyl)prop-2-en-1-ylidene]amino}butyl)amino]propyl}prop-2-enimidate | HMDB | | N1-Caffeoyl-N10-feruloylspermidine | HMDB | | Caffeoylferuloylspermidine | HMDB |
|
|---|
| Chemical Formula | C26H33N3O6 |
|---|
| Average Molecular Mass | 483.557 g/mol |
|---|
| Monoisotopic Mass | 483.237 g/mol |
|---|
| CAS Registry Number | 124935-81-5 |
|---|
| IUPAC Name | (2E)-3-(3,4-dihydroxyphenyl)-N-[3-({4-[(2E)-3-(4-hydroxy-3-methoxyphenyl)prop-2-enamido]butyl}amino)propyl]prop-2-enamide |
|---|
| Traditional Name | (2E)-3-(3,4-dihydroxyphenyl)-N-[3-({4-[(2E)-3-(4-hydroxy-3-methoxyphenyl)prop-2-enamido]butyl}amino)propyl]prop-2-enamide |
|---|
| SMILES | COC1=C(O)C=CC(\C=C\C(=O)NCCCCNCCCNC(=O)\C=C\C2=CC(O)=C(O)C=C2)=C1 |
|---|
| InChI Identifier | InChI=1S/C26H33N3O6/c1-35-24-18-20(6-10-22(24)31)8-12-25(33)28-15-3-2-13-27-14-4-16-29-26(34)11-7-19-5-9-21(30)23(32)17-19/h5-12,17-18,27,30-32H,2-4,13-16H2,1H3,(H,28,33)(H,29,34)/b11-7+,12-8+ |
|---|
| InChI Key | YUUGXIVVDZSRNR-MKICQXMISA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as hydroxycinnamic acids and derivatives. Hydroxycinnamic acids and derivatives are compounds containing an cinnamic acid (or a derivative thereof) where the benzene ring is hydroxylated. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Phenylpropanoids and polyketides |
|---|
| Class | Cinnamic acids and derivatives |
|---|
| Sub Class | Hydroxycinnamic acids and derivatives |
|---|
| Direct Parent | Hydroxycinnamic acids and derivatives |
|---|
| Alternative Parents | |
|---|
| Substituents | - Cinnamic acid amide
- Hydroxycinnamic acid or derivatives
- Methoxyphenol
- Phenoxy compound
- Catechol
- Phenol ether
- Styrene
- Methoxybenzene
- Anisole
- Alkyl aryl ether
- Phenol
- 1-hydroxy-4-unsubstituted benzenoid
- 1-hydroxy-2-unsubstituted benzenoid
- Benzenoid
- Monocyclic benzene moiety
- Secondary carboxylic acid amide
- Carboxamide group
- Amino acid or derivatives
- Secondary amine
- Ether
- Carboxylic acid derivative
- Secondary aliphatic amine
- Organic nitrogen compound
- Organonitrogen compound
- Organooxygen compound
- Hydrocarbon derivative
- Carbonyl group
- Organic oxide
- Organopnictogen compound
- Organic oxygen compound
- Amine
- Aromatic homomonocyclic compound
|
|---|
| Molecular Framework | Aromatic homomonocyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-03fr-1790000000-027fc5d47cf079ce5e59 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (2 TMS) - 70eV, Positive | splash10-01p9-1094002000-7e37738777675d8af40f | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-0ac3-0529300000-b07fac77f176021e1535 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0006-1912000000-0c716f66840feaab0245 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-002o-1910000000-bb85170fffbe2f8be233 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-001i-0411900000-7844e4d68321ecaf295a | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-01u3-0913400000-8425b0848c18f9b17db7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-002f-4900000000-7196e05b9becc2b01bc3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-001i-0000900000-558f7fbae7f469b7ebc3 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-0fl0-0362900000-c2cdde63d2bc5b490b16 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-004s-0910000000-1226e6c7ae4f2636692b | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-001i-0100900000-69408d32b7304a97d10e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-008i-0902700000-d2d0e157b745fad80be5 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-001r-0611900000-de76ea519fd7cdc9bf6e | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0033470 |
|---|
| FooDB ID | FDB011512 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | Not Available |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | 30777008 |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 131751431 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|