| Record Information |
|---|
| Version | 1.0 |
|---|
| Creation Date | 2016-05-25 23:50:25 UTC |
|---|
| Update Date | 2016-11-09 01:18:29 UTC |
|---|
| Accession Number | CHEM027596 |
|---|
| Identification |
|---|
| Common Name | Norisodomesticine |
|---|
| Class | Small Molecule |
|---|
| Description | Alkaloid from the leaves Laurus nobilis (bay laurel). Norisodomesticine is found in tea, sweet bay, and herbs and spices. |
|---|
| Contaminant Sources | |
|---|
| Contaminant Type | Not Available |
|---|
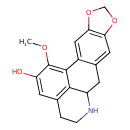
| Chemical Structure | |
|---|
| Synonyms | | Value | Source |
|---|
| 2-Hydroxy-1-methoxy-9,10-methylenedioxynoraporphine | HMDB | | Norjuzipjine | HMDB |
|
|---|
| Chemical Formula | C18H17NO4 |
|---|
| Average Molecular Mass | 311.332 g/mol |
|---|
| Monoisotopic Mass | 311.116 g/mol |
|---|
| CAS Registry Number | 80151-84-4 |
|---|
| IUPAC Name | 19-methoxy-5,7-dioxa-13-azapentacyclo[10.7.1.0²,¹⁰.0⁴,⁸.0¹⁶,²⁰]icosa-1(19),2,4(8),9,16(20),17-hexaen-18-ol |
|---|
| Traditional Name | 19-methoxy-5,7-dioxa-13-azapentacyclo[10.7.1.0²,¹⁰.0⁴,⁸.0¹⁶,²⁰]icosa-1(19),2,4(8),9,16(20),17-hexaen-18-ol |
|---|
| SMILES | COC1=C2C3=CC4=C(OCO4)C=C3CC3NCCC(C=C1O)=C23 |
|---|
| InChI Identifier | InChI=1S/C18H17NO4/c1-21-18-13(20)5-9-2-3-19-12-4-10-6-14-15(23-8-22-14)7-11(10)17(18)16(9)12/h5-7,12,19-20H,2-4,8H2,1H3 |
|---|
| InChI Key | GUVKEPNWVHYXGH-UHFFFAOYSA-N |
|---|
| Chemical Taxonomy |
|---|
| Description | belongs to the class of organic compounds known as aporphines. These are quinoline alkaloids containing the dibenzo[de,g]quinoline ring system or a dehydrogenated derivative thereof. |
|---|
| Kingdom | Organic compounds |
|---|
| Super Class | Alkaloids and derivatives |
|---|
| Class | Aporphines |
|---|
| Sub Class | Not Available |
|---|
| Direct Parent | Aporphines |
|---|
| Alternative Parents | Not Available |
|---|
| Substituents | Not Available |
|---|
| Molecular Framework | Aromatic heteropolycyclic compounds |
|---|
| External Descriptors | Not Available |
|---|
| Biological Properties |
|---|
| Status | Detected and Not Quantified |
|---|
| Origin | Not Available |
|---|
| Cellular Locations | Not Available |
|---|
| Biofluid Locations | Not Available |
|---|
| Tissue Locations | Not Available |
|---|
| Pathways | Not Available |
|---|
| Applications | Not Available |
|---|
| Biological Roles | Not Available |
|---|
| Chemical Roles | Not Available |
|---|
| Physical Properties |
|---|
| State | Not Available |
|---|
| Appearance | Not Available |
|---|
| Experimental Properties | | Property | Value |
|---|
| Melting Point | Not Available | | Boiling Point | Not Available | | Solubility | Not Available |
|
|---|
| Predicted Properties | |
|---|
| Spectra |
|---|
| Spectra | | Spectrum Type | Description | Splash Key | View |
|---|
| Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | splash10-003s-0090000000-4a236ae276ce4257db60 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (1 TMS) - 70eV, Positive | splash10-00xr-5039000000-b6ca4e65cc5844fd83c6 | Spectrum | | Predicted GC-MS | Predicted GC-MS Spectrum - GC-MS (Non-derivatized) - 70eV, Positive | Not Available | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-03di-0029000000-ae3a8ed498cf583ca171 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-03di-0096000000-d1f26ca68ece83a9fe28 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-0h2o-0390000000-1162f5ebf3635336d617 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03di-0019000000-338dbb83bb78c235c6c7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-03di-0069000000-4d75d6af24e219a9fb2f | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-01p6-1090000000-536cbc24552b218b1662 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Negative | splash10-03di-0009000000-43e89ef889a2eb4f124e | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Negative | splash10-03di-0019000000-c69dcebaacc3dea02a09 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Negative | splash10-0bti-0092000000-9270e39f37625d45e656 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 10V, Positive | splash10-03di-0009000000-4631b5d1b4d93faf70b7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 20V, Positive | splash10-03di-0009000000-4631b5d1b4d93faf70b7 | Spectrum | | Predicted LC-MS/MS | Predicted LC-MS/MS Spectrum - 40V, Positive | splash10-02ai-0090000000-419bc05173dc961931f4 | Spectrum |
|
|---|
| Toxicity Profile |
|---|
| Route of Exposure | Not Available |
|---|
| Mechanism of Toxicity | Not Available |
|---|
| Metabolism | Not Available |
|---|
| Toxicity Values | Not Available |
|---|
| Lethal Dose | Not Available |
|---|
| Carcinogenicity (IARC Classification) | Not Available |
|---|
| Uses/Sources | Not Available |
|---|
| Minimum Risk Level | Not Available |
|---|
| Health Effects | Not Available |
|---|
| Symptoms | Not Available |
|---|
| Treatment | Not Available |
|---|
| Concentrations |
|---|
| Not Available |
|---|
| External Links |
|---|
| DrugBank ID | Not Available |
|---|
| HMDB ID | HMDB0033361 |
|---|
| FooDB ID | FDB011389 |
|---|
| Phenol Explorer ID | Not Available |
|---|
| KNApSAcK ID | C00028723 |
|---|
| BiGG ID | Not Available |
|---|
| BioCyc ID | Not Available |
|---|
| METLIN ID | Not Available |
|---|
| PDB ID | Not Available |
|---|
| Wikipedia Link | Not Available |
|---|
| Chemspider ID | Not Available |
|---|
| ChEBI ID | Not Available |
|---|
| PubChem Compound ID | 69406100 |
|---|
| Kegg Compound ID | Not Available |
|---|
| YMDB ID | Not Available |
|---|
| ECMDB ID | Not Available |
|---|
| References |
|---|
| Synthesis Reference | Not Available |
|---|
| MSDS | Not Available |
|---|
| General References | |
|---|